b Department of Microbial Technology, School of Biological Sciences, Madurai Kamaraj University, Madurai 625021, India
Microbial infections are a growing problem in contemporary medicine. According to statistical evidence provided by WHO, many of the drug treatment breakthroughs of the last century could be lost through the spread of antimicrobial resistance [1]. For instance,Staphylococcus aureus,Pseudomonas aeruginosa and Klebsiella pneumoniae are important pathogens causing invasive diseases such as sepsis,meningitis,necrotizing fasciitis,pneumonia [2],nosocomial pneumonia [3],cystic fibrosis,acute leukemia, organ transplants,and intravenous-drug addiction [4]. Some of these pathogens have been reported to develop resistance [5] to the well-known commercially available drugs. As a result,many infectious diseases may one day become uncontrollable and could rapidly spread throughout the world. Consequently,the discovery of potent antibiotic drugs is considered to be one of the greatest scientific and medical goals.
There is a growing interest pertaining to the synthesis of bioactive heterocyclic compounds in pharmacy. Among various heterocyclic compounds,pyrimidine derivatives have apparently gained considerable importance owing to their varied biological activities such as adenosine receptor antagonist [6],anti-inflammatory [7],CDK inhibitor [8],calcium channel antagonist [9] and anti-tumor [10] activities. On the other hand,a promising diverse pharmacological activity is shown by the 1,2,3-triazole nucleus such as anti-biotic,anti-fungal [11],anti-cancer [12],HIV protease inhibitor [13] and chemotherapeutic activities [14].
In view of the bioactivity profiles of the individual heterocycles, the synthesis of hybrid molecules containing both of the above said moieties in a single frame was attempted. Thus,the present study was undertaken to synthesize and investigate the anti-microbial activities of a series of novel 1,2,3-triazolyl-2-aminopyrimidine hybrids against three human pathogenic bacteria viz. K. pneumoniae, S. aureus and P. aeruginosa,the details of which are presented vide infra. 2. Experimental
Typical procedure: A mixture of (E)-1-(1-benzyl-5-methyl-1H- 1,2,3-triazol-4-yl)-3-phenylprop-2-en-1-one,2a (0.2 g,0.65mmol), guanidine hydrochloride (0.18 g,1.88mmol) and NaOH (0.04 g, 1mmol) in water or ethanol (10 mL) was refluxed for 30-40 min. Then,the reactionmixture was poured onto excess crushed ice and neutralized with dilute hydrochloric acid. The precipitated 1,2,3- triazolyl-2-aminopyrimidine (3a) was filtered and recrystallized from ethanol. Yield 0.19 g (85%) in ethanol,0.18 g (82%) in water.
4-(1-Benzyl-5-methyl-1H-1,2,3-triazol-4-yl)-6-phenylpyrimidin- 2-amine (3a): Obtained as white solid in 82% yield,mp 152°C; IR (KBr,cm-1): υ 3411,3192,1677,1552,1462,1355,1181,834, 729,700; 1H NMR (300 MHz,CDCl3): δ 8.12-8.09 (m,2 H,ArH), 7.98 (s,1H,55CH),7.49-7.18 (m,8H,ArH),5.56 (s,2H,C6H5-CH2), 5.07 (brs,2H,NH2),2.62 (s,3H,CH3); 13C NMR (75 MHz,CDCl3): δ 165.87,163.14,160.56,142.30,137.57,134.51,134.00,133.94, 130.48,130.42,129.06,128.71,128.66,128.41,127.17,104.50, 51.80,9.82. MS (ESI): m/z 343.33 (M+H); Anal. Calcd. for CH3H18N6: C,70.16; H,5.30; N,24.54; Found: C,70.09; H,5.32; N,24.50.
By a similar procedure,4-(1-benzyl-5-methyl-1H-1,2,3-triazol- 4-yl)-6-arylpyrimidin-2-amines (3b-l) were synthesized. Their characterization data and copies of NMR (1D and 2D),FT-IR and LC-MS spectra of representative compounds can be found in Supporting information. 3. Results and discussion
Over the past decades,many protocols have been developed for the synthesis of pyrimidine derivatives [15],and most of these involve the use of hazardous organic solvents [16, 17]. Hence,ecofriendly organic reactions that occur under solvent-free conditions or use water have attained much importance and are of current interest. In particular,as a reaction medium,water offers many practical and economic advantages including low cost,environmental compatibility,and safety among all available solvents,thus leading to environmentally-friendly chemical processes [18]. In view of this,recently we have accomplished the synthesis of amides and various heterocycles/hybrid heterocycles under solvent-free/water medium conditions [19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30].
Perusal of literature suggests that there are no reports on the synthesis of 1,2,3-triazolyl-2-aminopyrimidine hybrids. In connection with our goal to conduct organic reactions in water medium and in continuation of our earlier report on the water promoted synthesis of 1,2,3-triazolyl-pyrimidine-2-thione hybrids [28] as a highly potent antibacterial agent,we hereby submit the first report on the eco-friendly synthesis of 1,2,3-triazolyl-2- aminopyrimidine hybrids and their anti-bacterial activity.
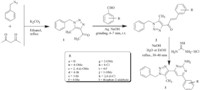
At the outset,synthesis of 1,2,3-triazolyl-2-aminopyrimidine (3a) was attempted while varying the bases (Scheme 1 and Table 1),i.e. a mixture of 1,2,3-triazolyl chalcone [26] (2a,1 equiv.), guanidine hydrochloride (3 equiv) and base (1.5 equiv) was refluxed in ethanol or water (10 mL). Completion of the reaction was monitored by TLC. It is noted that basicity is one of the key points behind the success of the reaction (Table 1). Moreover, water seems to be the proper solvent to fulfill the reaction by achieving the best solubility of the salts viz. guanidine hydrochloride and sodium hydroxide,thus promoting the reaction. The yields in ethanol and water were comparable.

|
Download:
|
| Scheme 1.Optimization for the synthesis of 1,2,3-triazolyl-2-aminopyrimidine hybrids (3a). | |
| Table 1 Optimization for the synthesis of 1,2,3-triazolyl-2-aminopyrimidine hybrids (3a). |
All twelve compounds exhibit different levels of inhibition against the previously mentioned human pathogens (both Grampositive and Gram-negative) (Scheme 2 and Table 2). Among them, compounds coded 3a (-H),3b (-4OMe),3c (-2,4 diOMe),3g (- 2OMe),3i (-4F),3k (-2,4 diCl) (MIC 2 mg/mL) and 3d (-4Br) (MIC 5 mg/mL) were more potent against P. aeruginosa than tetracycline (MIC 10 mg/mL). Meanwhile,the compounds 3j (-3OMe) and 3l (2- thiophenyl) show equal inhibition (MIC 10 mg/mL) to tetracycline. However,the rest of the compounds showed lower levels of inhibition (MIC 25-35 mg/mL). In the case of Klebsiella pneumonia, compounds 3b and 3c were equally potent (MIC 2 mg/mL) as tetracycline. The compounds 3a and 3e (-3Br) exhibited moderate potency (MIC 5 mg/mL) and the others showed less potency (MIC > 5 mg/mL). On the other hand,all the compounds exhibited less potency (MIC ≥ 30 mg/mL) toward S. aureus than tetracycline (MIC 15 mg/mL). This may be attributed to the thick peptidoglycan ayer of the cell wall of Gram positive S. aureus. Also,the binding of the cell-surface proteins of the Gram positive S. aureus might have inactivated the compounds. The above results project 3b and 3c as highly potent compounds with remarkable antibacterial activity in comparison to the commercial control.
|
Download:
|
| Scheme 2.Synthesis of 1,2,3-triazolyl-2-aminopyrimidine hybrids (3). | |
| Table 2 Synthesis of 1,2,3-triazolyl-2-aminopyrimidine hybrids (3a-l)a in ethanol and water and their in vitro anti-bacterial activity against three human pathogens. |
Structure-activity relationship (SAR) in these compounds demonstrated that compounds 3b and 3c with substituents viz. -4OMe and -2,4 diOMe in the phenyl ring attached to the C-6 carbon of 2-amino pyrimidine moiety are more potent against all the tested pathogens. Subsequently,various substituents were introduced in either of the -ortho,-meta or -para position of the phenyl ring to evaluate their effect on biological activity. Their order of activity compared to compound (3a) with the unsubstituted phenyl ring linked to C-6 carbon of 2-amino pyrimidine is in the order -4OMe ≥ -2,4-diOMe > -H > -4Br > -2OMe > -4F > -2,4 diCl > -3OMe > -thiophene > -3Br. However,compounds 3f with -4Me and 3h with -4Cl substituents did not show notable activity against all the tested pathogens. 4. Conclusion
In conclusion,an elegant,environmentally-friendly water promoted synthesis of a library of 1,2,3-triazolyl-2-aminopyrimidine hybrids has been achieved. All the synthesized compounds were evaluated for antibacterial activity. Many of them exhibited potent activity,and some of them were equally potent or of higher potency than the commercially marketed tetracycline. Acknowledgments
The authors thank the UGC (BSR) and DST-PURSE for financial support and Department of Science and Technology,New Delhi, India for funds under the IRHPA program for the purchase of a NMR spectrometer. Appendix A. Supplementary data
Supplementary data associated with this article can be found,in the online version,at http://dx.doi.org/10.1016/j.cclet.2013.12.017.
| [1] | R.P. Mishra, E. Oviedo-Orta, P. Prachi, R. Rappuoli, F. Bagnoli, Vaccines and antibiotic resistance, Curr. Opin. Microbiol. 15 (2012) 596-602. |
| [2] | L.K. Siu, K.M. Yeh, J.C. Lin, C.P. Fung, F.Y. Chang, Klebsiella pneumoniae liver abscess: a new invasive syndrome, Lancet Infect. Dis. 12 (2012) 881-887. |
| [3] | A.R. Marra, S.B. Wey, A. Castelo, et al., Nosocomial bloodstream infections caused by Klebsiella pneumoniae: impact of extended-spectrum beta-lactamase (ESBL) production on clinical outcome in a hospital with high ESBL prevalence, BMC Infect. Dis. 6 (2006) 24, http://dx.doi.org/10.1186/1471-2334-6-24. |
| [4] | (a) H. Harikrishnan, A. Naif AbdullahI, K. Ponmurugan, R. Shyam Kumar, Nanocomposite using Saccharomyces cerevisiae and its antimicrobial activity against pathogens causing nosocomial, Chalcogenide Lett. 9 (2012) 509-515; (b) G.P. Bodey, R. Bolivar, V. Fainstein, L. Jadeja, Infections Caused by Pseudomonas aeruginosa, Rev. Infect. Dis. 5 (1983) 279-313. |
| [5] | I. Chopra, M. Roberts, Tetracycline antibiotics: mode of action, applications, molecular biology, and epidemiology of bacterial resistance, Microbiol. Mol. Biol. Rev. 65 (2001) 232-260. |
| [6] | V. Yaziji, D. Rodríguez, H. Gutié rrez-de-Terán, et al., Pyrimidine derivatives as potent and selective A3 adenosine receptor antagonists, J. Med. Chem. 54 (2011) 457-471. |
| [7] | M.S. Mohamed, S.M. Awad, A.I. Sayed, Synthesis of certain pyrimidine derivatives as antimicrobial agents and anti-inflammatory agents, Molecules 15 (2010) 1882-1890. |
| [8] | X. Chu, W. Depinto, D. Bartkovitz, et al., Discovery of [4-amino-2-(1-methanesulfonylpiperidin-4-ylamino)pyrimidin-5-yl](2,3-difluoro-6-methoxyphenyl) methanone (R547) A potent and selective cyclin-dependent kinase inhibitor with significant in vivo antitumor activity, J. Med. Chem. 49 (2006) 6549-6560. |
| [9] | K.S. Atwal, G.C. Rovnyak, S.D. Kimball, et al., Dihydropyrimidine calcium channel blockers. Ⅱ. 3-Substituted-4-aryl-1,4-dihydro-6-methyl-5-pyrimidinecarboxylic acid esters as potentmimics of dihydropyridines, J.Med. Chem. 33 (1990) 2629-2635. |
| [10] | (a) N. Singh, S.K. Pandey, N. Anand, et al., Synthesis, molecular modeling and bioevaluation of cycloalkyl fused 2-aminopyrimidines as antitubercular and antidiabetic agents, Bioorg. Med. Chem. 21 (2011) 4404-4408; (b) Y.F. Zhao, Z.J. Liu, X. Zhai, et al., Synthesis and in vitro antitumor activity of novel diaryl urea derivatives, Chin. Chem. Lett. 24 (2013) 386-388. |
| [11] | X.L. Wang, K. Wan, C.H. Zhou, Synthesis of novel sulfanilamide-derived 1,2,3-triazoles and their evaluation for antibacterial and antifungal activities, Eur. J. Med. Chem. 45 (2010) 4631-4639. |
| [12] | S.R. Wang, Q.L. Wang, Y. Wang, et al., Novel anthraquinone derivatives: synthesis via click chemistry approach and their induction of apoptosis in BGC gastric cancer cells via reactive oxygen species (ROS)-dependent mitochondrial pathway, Bioorg. Med. Chem. 18 (2008) 6505-6508. |
| [13] | M. Whiting, J.C. Tripp, Y.C. Lin, et al., Rapid discovery and structure-activity profiling of novel inhibitors of human immunodeficiency virus type 1 protease enabled by the copper(I)-catalyzed synthesis of 1,2,3-triazoles and their further functionalization, J. Med. Chem. 49 (2006) 7697-7710. |
| [14] | P. Pramitha, D. Bahulayan, Stereoselective synthesis of bio-hybrid amphiphiles of coumarin derivatives by Ugi-Mannich triazole randomization using copper catalyzed alkyne azide click chemistry, Bioorg. Med. Chem. Lett. 22 (2012) 2598-2603. |
| [15] | (a) M. Yu, S.S. Pochapsky, B.B. Snider, Synthesis of 7-epineoptilocaulin, mirabilin B, and isoptilocaulin. A unified biosynthetic proposal for the ptilocaulin and batzelladine alkaloids. Synthesis and structure revision of netamines E and G, J. Org. Chem. 73 (2008) 9065-9074; (b) A. Rahmati, Z. Khalesi, Catalyst free synthesis of fused pyrido[2,3-d]pyrimidines and pyrazolo[3,4-b]pyridines in water, Chin. Chem. Lett. 23 (2012) 1149-1152. |
| [16] | N. Sunduru, S. Nishi, P.M.S. Palne, S. Chauhan, Gupta Synthesis and antileishmanial activity of novel 2,4,6-trisubstituted pyrimidines and 1,3,5-triazines, Eur. J. Med. Chem. 44 (2009) 2473-2481. |
| [17] | M. Meisenbach, T. Allmendinger, C.P. Mak, Scale-up of the synthesis of a pyrimidine derivative directly on solid support, Org. Process Res. Dev. 7 (2003) 553-558. |
| [18] | C.I. Herrerias, X.Q. Yao, Z.P. Li, C.J. Li, Reactions of C-H bonds in water, Chem. Rev. 107 (2007) 2546-2562. |
| [19] | M. Sathishkumar, P. Shanmugavelan, S. Nagarajan, et al., Solvent-free protocol for amide bond formation via trapping of nascent phosphazenes with carboxylic acids, Tetrahedron Lett. 52 (2011) 2830-2833. |
| [20] | S. Nagarajan, P. Ran, P. Shanmugavelan, et al., The catalytic activity of titania nanostructures in the synthesis of amides under solvent-free conditions, New J. Chem. 36 (2012) 1312-1319. |
| [21] | S. Nagarajan, P. Shanmugavelan, M. Sathishkumar, et al., Chemoselectivity in coupling of azides with thioacids in solution-phase and solvent-free conditions, Synth. Commun. 43 (2013) 37-41. |
| [22] | M. Sathishkumar, K. Palanikumar, A. Mariappan, S. Archana, A. Ponnuswamy, An environmentally benign solvent/catalyst-free one-pot synthesis of N-substituted phthalimides via Aza-wittig reaction, J. Iran. Chem. Soc. 9 (2012) 681-685. |
| [23] | P. Shanmugavelan, S. Nagarajan, M. Sathishkumar, et al., Efficient synthesis and in vitro antitubercular activity of 1,2,3-triazoles as inhibitors of Mycobacterium tuberculosis, Bioorg. Med. Chem. Lett. 21 (2011) 7273-7276. |
| [24] | P. Shanmugavelan, M. Sathishkumar, S. Nagarajan, et al., The first solvent-free, microwave-accelerated, three-component synthesis of thiazolidin-4-ones via one-pot tandem Staudinger/aza-Wittig reaction, J. Heterocyclic. Chem. (1705), http://dx.doi.org/10.1002/jhet. |
| [25] | A. Ponnuswamy, P. Shanmugavelan, S. Nagarajan, M. Sathishkumar, The first onepot, solvent-free, microwave-accelerated, three-component synthesis of spirothiazolidin-4-ones via Staudinger/Aza-Wittig coupling/cyclization, Helv. Chim. Acta 95 (2012) 922-928. |
| [26] | P. Shanmugavelan, M. Sathishkumar, S. Nagarajan, A. Ponnuswamy, A facile synthesis of 1,2,3-triazolyl indole hybrids via SbCl3-catalysed Michael addition of indoles to 1,2,3-triazolyl chalcones, J. Chem. Sci. 124 (2012) 941-950. |
| [27] | M. Sathishkumar, P. Shanmugavelan, S. Nagarajan, M. Dinesh, A. Ponnuswamy, Water promoted one pot three-component synthesis of tetrazoles, New J. Chem. 37 (2013) 488-493. |
| [28] | N. Sangaraiah, S. Murugan, S. Poovan, et al., Facile water promoted synthesis of 1,2,3-triazolyl dihydropyrimidine-2-thione hybrids—highly potent antibacterial agents, Eur. J. Med. Chem. 58 (2012) 464-469. |
| [29] | P. Shanmugavelan, S. Nagarajan, et al., An efficient and environmentally benign access towards synthesis of novel 1,2,3-triazolyl-pyrazoline hybrids, Lett. Org. Chem., in press. |
| [30] | P. Shanmugavelan, M. Sathishkumar, S. Nagarajan, A. Ponnuswamy, An efficient and facile synthesis of novel 1,2,3-triazolyl-N-acylpyrazoline hybrids, Chin. Chem. Lett. 25 (2014) 146-148. |