Chemical composition and alteration assemblages of eudialyte in the Saima alkaline complex, Liaoning Province, and its implication for alkaline magmatic-hydrothermal evolution
碱性岩通常是指硅酸不饱和、富碱的一类岩石,其来源于富集岩石圈地幔,多形成于大陆裂谷、板内和造山带后伸展环境(任康绪, 2003; Sørensen, 1997; Zhu et al., 2017)。因其来源的特殊性,碱性岩研究常涉及到岩石圈地幔演化、壳幔相互作用、地幔流体交代等众多固体地球科学的前沿问题(Markl et al., 2010; Zhu et al., 2017);此外碱性岩常富含多种碱性矿物,并伴生许多重要的稀有金属矿物和稀土矿物,如铈铌钙钛矿、氟碳铈矿、异性石等(牛贺才和林传仙, 1994; Mitchell and Chakhmouradian, 1996; Schilling et al., 2011)。碱性岩矿物学研究不但为揭示碱性岩浆-热液演化规律提供重要信息,而且在探究碱性岩型稀有、稀土金属矿化机制方面起到重要作用。异性石[Na4(Ca, Ce)2(Fe2+, Mn2+)ZrSi8O22(OH, Cl)2]是碱性岩的标志性副矿物之一,最早发现于南格陵兰Ilímaussaq碱性岩体(Stromeyer, 1819; Johnsen and Grice, 1999),广泛分布于俄罗斯科拉半岛Khibiny、Lovozero、南非Pilanesberg和我国辽宁赛马等碱性岩体的岩浆、热液阶段(陈肇博等, 1996; Mitchell and Chakhmouradian, 1996; Olivo and Williams-Jones, 1999; Schilling et al., 2011)。异性石化学成分受结晶环境影响明显,且容易被晚期热液流体交代,是查明碱性岩浆来源、指示岩浆结晶分异程度、探究晚期热液流体性质、高场强元素热液活动性的理想矿物之一(Salvi et al., 2000; Karup-Møller et al., 2010; Wu et al., 2010; Schilling et al., 2011; Chakrabarty et al., 2012; Karup-Møller and Rose-Hansen, 2013)。
辽宁赛马碱性岩体位于我国华北克拉通东北缘辽东半岛北部宽甸地区,早期以产碱性岩型铀矿而被地质工作者所关注(北京铀矿地质研究所赛马矿床研究组, 1977; 陈肇博等, 1996)。长期以来,国内许多学者对赛马碱性岩体的基础矿物学特征(彭琪瑞等, 1962; 陈肇博等, 1996; Yang et al., 2012)、岩石地球化学特征(周玲棣和王扬传, 1991; 谭东娟等, 1998)、成岩成矿年代和模式(景立珍等, 1995; 陈肇博等, 1996; 周玲棣等, 1996)、矿床地质特征(北京铀矿地质研究所赛马矿床研究组, 1977; 陈肇博等, 1996)等方面进行了大量的研究,并取得了一系列的成果。近年来随着研究理论的进步和实验方法提升,部分学者对赛马碱性岩体的形成年代、物质来源、成因模式和矿物特征等方面进行了更精细的限定和研究(Wu et al., 2010, 2015, 2016; Zhu et al., 2016, 2017)。但对该岩体晚期岩浆演化特征、热液流体性质、来源及流体作用下稀有稀土元素地球化学行为等方面缺乏系统的研究,很大程度上制约了对该岩体岩浆-热液演化过程的认识。
本文在已有研究及详细的野外地质观察、岩相学研究的基础上,通过对辽宁赛马岩体不同阶段异性石化学成分特征和蚀变矿物组合进行详细的观察和研究,揭示该岩体晚期热液流体的性质及作用,并在此基础上进一步探讨稀有稀土元素热液活动性,以期对碱性岩体岩浆-热液演化和稀有稀土元素矿化机制提供进一步的认识。
1 地质背景 赛马碱性岩体隶属于赛马碱性杂岩区,也称凤城碱性杂岩区,地理位置上分布于E120°11′~124°48′、N40°52′~41°5′之间的辽宁省凤城市赛马镇东部。构造意义上,该杂岩区出露于辽北地块南缘,南部为辽河群广泛出露的辽吉造山带,即传统地质学所指的太子河-浑江古凹陷南侧,营口-宽甸隆起北侧。杂岩区呈东西向展布,出露面积约280km2,主要分为东部的柏林川岩体、中部的顾家、叆阳岩体和西部的赛马岩体,分别侵位于辽河群、青白口系钓鱼台组和南芬组、震旦系桥头组、寒武系、奥陶系,而被下侏罗统长梁子组不整合覆盖(陈肇博等, 1996; Zhu et al., 2016)。
赛马碱性岩体位于凤城碱性杂岩区的西部,出露面积约20km2,岩性组成复杂,主要分为第一期主体霞石正长岩(70%),中部响岩(20%)和第二期异霞正长岩(5%,图 1)。岩体北部、西部和南部的围岩为元古界辽河群白云质大理岩,西南部为辽河群千枚岩,东北部为震旦系石英岩和寒武系灰岩,东部被侏罗系砂砾岩和煤系地层所覆盖(陈肇博等, 1996)。其中异霞正长岩是赛马铀、钍、稀土矿床的主要含矿母岩,矿石矿物包括绿层硅铈钛矿、异性石、沥青铀矿、铀方钍矿等(陈肇博等, 1996)。最新的岩石地球化学及锆石Hf-O同位素证据认为赛马碱性岩体可能是再循环的古老陆壳产生的熔体或流体交代大陆岩石圈地幔的产物(Zhu et al., 2017),而锆石、异性石U-Pb年龄显示赛马碱性岩体的形成年龄约为224~230Ma(Wu et al., 2010; Zhu et al., 2016)。
2 样品描述 异性石赋存于赛马碱性岩体异霞正长岩中,据前人资料显示此类岩石在地表出露较少,多发育于河床位置或岩体深部(陈肇博等, 1996)。我们此次采样点位于岩体西北角山顶涓卧线292高压线塔附近,坐标为E124°16′27.8″、N40°58′20.6″,草绿色异霞正长岩侵入早期粗粒霞石正长岩。
异霞正长岩整体呈草绿色,具中粒至中粗粒半自形全晶质粒状结构和块状构造(图 2a),主要矿物包括霞石(20%~25%)、钾长石(25%~30%)、霓石(30%~40%)、异性石(5%~10%)、榍石(5%)和少量黑云母、闪叶石,矿物局部分布不均匀。霞石和钾长石是异霞正长岩主要的浅色矿物,粒径2~10mm不等,钾长石镜下呈他形至半自形粒状,表面干净。霞石手标本中呈肉红色,他形至半自形粒状晶体,与钾长石、霓石等造岩矿物共生(图 2a),背散射电子镜下霞石颗粒大多被钠沸石交代。异霞正长岩中绿色霓石多呈粒状或细针状、毛发状集合体形式产出(图 2b)。手标本中异性石为淡红色,半自形粒状,约为0.1~1mm,在岩石中呈稀散不均匀分布,有时呈集合体产出。薄片中异性石呈无色透明,或略带淡粉色,正突起中等,表面常见裂纹,部分异性石发生蚀变(图 2b)。异性石常与草绿色霓石紧密共生或包裹后者,说明异性石的形成不早于霓石。副矿物包括方钍石、菱锶矿、铈铌钙钛矿、层硅铈钛矿、何作霖矿和闪叶石等。
3 样品处理及分析方法 选择新鲜的异霞正长岩磨成光薄片进行背散射图像观察以及电子探针成分分析。矿物背散射电子像观察主要在东华理工大学省部共建核资源与环境国家重点实验室培育基地利用FEI-Nova NanoSEM40场发射扫描电镜完成,矿物化学成分定量分析和部分背散射电子像观察在南京大学内生金属矿床成矿机制研究国家重点实验室利用JEOL-JXA8100电子探针完成,异性石假晶元素特征X射线面分析在东华理工大学省部共建核资源与环境国家重点实验室培育基地利用同型号的电子探针完成。仪器工作条件如下:工作电压15kV,加速电流20nA,为了防止钠在测试过程中挥发,根据异性石、钠锆石和部分霓石的大小,电子束束斑直径调整为10μm或20μm,其余矿物测量束斑直径2μm。标准样品选用萤石(F)、硅铍铝钠石(Cl)、硬玉(Na、Si)、透长石(K)、斜长石(Ca、Al)、黑云母(Fe、Mg)、锆石(Zr)、金属铪(Hf)、红钛锰矿(Ti、Mn)、铌酸锶钡(Nb、Sr、Ba)、晶质铀矿(U、Th、Pb)、独居石(P、La、Ce、Pr、Nd、Sm)和部分稀土磷酸盐(Eu、Gd、Tb、Dy、Ho、Er、Tm、Yb、Y)。Si、Al、Mg、Ca、Fe、Na、K、F、Cl等主量元素或易挥发元素特征峰测量时间为10s,背景测量时间为5s;P、Nb、Ti、Zr、Hf、U、Th、Cr、Y、Mn、Pb、Sr、Ba和REEs等微量元素特征峰测量时间为20s,背景测量时间为10s,所有测试数据均采用ZAF程序进行了校正处理。
4 分析结果 赛马碱性岩体响岩、霞石正长岩、异霞正长岩全岩主微量地球化学数据及岩浆期主要矿物类型、化学成分等可能涉及本文讨论的内容见文献(Wu et al., 2015, 2016)。总体而言,异霞正长岩体系富碱、富钠、富流体挥发分和低氧逸度的特点,是异性石、层硅铈钛矿等锆矿物、稀土矿物相对富集的主要原因(Wu et al., 2016)。
4.1 原生异性石 4.1.1 岩浆早期异性石 背散射电子像下,早期异性石呈自形或半自形粒状,粒径0.1~1mm不等,表面裂纹发育明显,部分呈粒状集合体形式产出,与钾长石、霞石和霓石等早期造岩矿物共生(图 2c),从图 2b中异性石和霓石的关系,可得知异性石的结晶不早于霓石。少数异性石颗粒具有明显的成分环带特征,即异性石形成过程中成分差异引起核部较暗,边部较亮的现象(图 2d)。
具环带特征的异性石核部化学成分分布较为集中,总量接近100%,并具有较高的Fe(5.92%~6.80% FeOT)、Si(49.40%~54.83% SiO2)含量,较低的Mn(0.96%~1.41% MnO)、Sr(1.80%~2.38% SrO)、Nb(0.21%~0.97% Nb2O5)和REE(含Y,0.46%~0.98% REE2O3)含量(表 1、详见电子版附表 1)。相比而言,边部化学成分总量约为96%~99%,可能含有电子探针无法检测的OH-、CO32-等阴离子(Schilling et al., 2011)。此外边部具有较低的Fe(4.59%~6.13% FeOT)、Si(45.53%~52.16% SiO2)含量,较高的Mn(1.56%~2.37% MnO)、Sr(2.78%~4.38% SrO)、Nb(1.60%~3.71% Nb2O5)和REE(含Y,1.73%~4.66% REE2O3)含量。核部和边部具有类似的Cl(0.81%~1.42% Cl)含量(图 3)。
附表 1
Appendix Table 1
附表 1(Appendix Table 1)
附表 1
赛马岩体异霞正长岩各阶段异性石电子探针成分(wt%)
Appendix Table 1
Electron-microprobe results for different eudialyte from the lujavrite of the Saima complex (wt%)
| 产状 |
|
早期异性石核部 |
|
早期异性石边部 |
|
晚期异性石 |
| 序号 | |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
12 |
13 |
14 |
15 |
16 |
17 |
18 |
19 |
20 |
21 |
22 |
23 |
24 |
25 |
26 |
27 |
28 |
29 |
30 |
31 |
32 |
33 |
34 |
35 | |
36 |
37 |
38 |
39 |
40 |
41 |
42 |
43 |
44 |
45 |
46 |
47 |
48 |
49 |
50 |
51 |
52 |
53 |
54 |
55 |
56 |
57 |
58 |
59 |
60 |
61 |
62 | |
63 |
64 |
65 |
66 |
67 |
68 |
69 |
70 |
71 |
72 |
73 |
74 |
75 |
76 |
77 |
78 |
79 |
80 |
81 |
82 |
83 |
84 |
85 |
86 |
87 |
88 |
89 |
90 |
91 |
92 |
93 |
94 |
| Nb2O5 | |
0.97 |
0.44 |
0.62 |
0.35 |
0.27 |
0.21 |
0.46 |
0.26 |
0.64 |
0.82 |
0.72 |
0.72 |
0.70 |
0.84 |
0.94 |
0.72 |
0.89 |
0.75 |
0.62 |
0.60 |
0.80 |
0.68 |
0.69 |
0.59 |
0.71 |
0.58 |
0.90 |
0.73 |
0.72 |
0.81 |
0.47 |
0.46 |
0.66 |
0.53 |
0.67 | |
3.23 |
2.55 |
2.16 |
2.17 |
2.40 |
3.84 |
3.21 |
1.60 |
2.99 |
2.87 |
3.11 |
3.17 |
3.27 |
3.64 |
3.36 |
2.98 |
2.85 |
3.17 |
3.28 |
2.71 |
3.05 |
2.95 |
3.39 |
3.43 |
2.89 |
2.78 |
3.34 | |
3.12 |
2.55 |
3.31 |
4.02 |
3.38 |
3.35 |
3.59 |
3.63 |
2.69 |
2.82 |
3.61 |
3.01 |
3.36 |
3.03 |
3.60 |
3.75 |
3.27 |
3.14 |
3.34 |
3.26 |
3.29 |
2.76 |
3.35 |
2.73 |
2.91 |
2.47 |
2.99 |
3.33 |
3.53 |
2.49 |
3.36 |
3.12 |
| Ta2O5 |
0.04 |
0.23 |
— |
0.13 |
0.08 |
0.21 |
0.01 |
— |
0.19 |
0.02 |
— |
0.03 |
0.13 |
0.18 |
0.07 |
— |
— |
0.25 |
0.04 |
0.15 |
0.13 |
— |
0.05 |
— |
0.16 |
— |
0.10 |
— |
0.18 |
0.04 |
— |
— |
— |
— |
0.01 |
0.26 |
0.23 |
0.16 |
— |
0.05 |
0.12 |
— |
0.09 |
0.15 |
0.25 |
0.11 |
0.22 |
— |
— |
0.02 |
0.02 |
— |
0.27 |
— |
0.01 |
0.16 |
0.07 |
— |
0.05 |
0.29 |
0.04 |
0.35 |
0.21 |
0.24 |
0.15 |
— |
0.25 |
0.03 |
0.01 |
— |
0.19 |
— |
— |
0.01 |
0.24 |
0.21 |
0.10 |
0.12 |
— |
— |
— |
0.13 |
0.29 |
— |
0.15 |
— |
— |
— |
0.23 |
0.06 |
0.21 |
0.24 |
— |
0.03 |
| SiO2 |
53.74 |
54.62 |
53.80 |
54.83 |
54.60 |
54.59 |
54.80 |
53.59 |
53.96 |
53.76 |
53.68 |
49.59 |
53.86 |
53.61 |
53.18 |
53.42 |
53.39 |
49.40 |
50.47 |
53.64 |
52.75 |
53.17 |
53.20 |
53.88 |
52.08 |
53.82 |
52.69 |
53.36 |
52.35 |
52.55 |
53.64 |
54.04 |
50.83 |
50.76 |
53.21 |
45.31 |
50.45 |
48.01 |
48.58 |
49.77 |
48.13 |
47.49 |
50.60 |
47.14 |
47.53 |
46.56 |
46.44 |
46.62 |
45.53 |
45.66 |
46.38 |
48.60 |
45.65 |
46.27 |
48.98 |
47.74 |
46.50 |
46.19 |
49.15 |
50.30 |
50.82 |
50.01 |
45.47 |
46.11 |
46.13 |
49.71 |
49.25 |
47.65 |
49.73 |
47.88 |
49.54 |
48.27 |
48.53 |
48.10 |
49.40 |
50.39 |
49.77 |
49.60 |
48.93 |
49.22 |
48.90 |
50.08 |
49.06 |
47.44 |
49.52 |
49.64 |
50.02 |
49.34 |
49.31 |
49.53 |
48.83 |
49.31 |
50.10 |
45.63 |
| TiO2 |
0.43 |
0.90 |
0.34 |
0.51 |
0.50 |
0.81 |
0.61 |
0.76 |
0.59 |
0.45 |
0.38 |
0.29 |
0.39 |
0.38 |
0.40 |
0.38 |
0.34 |
0.44 |
0.34 |
0.34 |
0.39 |
0.40 |
0.44 |
0.41 |
0.36 |
0.48 |
0.43 |
0.45 |
0.39 |
0.39 |
0.42 |
0.46 |
0.35 |
0.38 |
0.37 |
0.09 |
0.25 |
0.29 |
0.22 |
0.25 |
0.11 |
0.12 |
0.36 |
0.20 |
0.12 |
0.14 |
0.10 |
0.13 |
0.10 |
0.11 |
0.20 |
0.25 |
0.14 |
0.10 |
0.23 |
0.14 |
0.19 |
0.16 |
0.13 |
0.22 |
0.26 |
0.18 |
0.40 |
0.68 |
0.34 |
0.28 |
0.39 |
0.57 |
0.43 |
0.42 |
0.94 |
0.75 |
0.45 |
0.62 |
0.36 |
0.48 |
0.16 |
0.16 |
0.63 |
0.69 |
0.38 |
0.19 |
0.67 |
0.53 |
0.50 |
0.76 |
0.47 |
0.64 |
0.49 |
0.44 |
0.43 |
0.94 |
0.50 |
0.32 |
| ZrO2 |
12.06 |
11.57 |
12.80 |
13.06 |
12.63 |
11.86 |
12.51 |
12.28 |
12.10 |
12.33 |
12.64 |
11.32 |
12.32 |
12.34 |
12.38 |
11.86 |
12.31 |
11.57 |
12.32 |
12.73 |
12.40 |
12.61 |
12.84 |
12.69 |
12.27 |
12.56 |
12.18 |
12.42 |
12.72 |
12.31 |
12.05 |
12.56 |
12.38 |
12.06 |
12.34 |
11.29 |
12.25 |
11.54 |
11.97 |
12.20 |
11.43 |
11.91 |
12.29 |
12.12 |
11.67 |
11.56 |
11.83 |
11.75 |
11.27 |
11.21 |
11.24 |
12.00 |
11.43 |
10.50 |
11.86 |
11.69 |
11.67 |
11.98 |
12.08 |
12.55 |
12.46 |
12.84 |
11.66 |
11.65 |
11.90 |
12.30 |
12.45 |
11.65 |
12.35 |
11.91 |
11.91 |
12.16 |
12.14 |
12.20 |
12.09 |
12.40 |
11.93 |
12.06 |
12.75 |
12.39 |
12.26 |
12.22 |
11.95 |
11.47 |
12.76 |
12.34 |
12.24 |
12.12 |
11.81 |
12.70 |
12.60 |
12.47 |
13.13 |
11.31 |
| HfO2 |
0.18 |
0.09 |
0.13 |
0.22 |
0.18 |
0.18 |
0.21 |
0.17 |
0.22 |
0.14 |
0.11 |
0.28 |
0.23 |
0.05 |
0.36 |
0.21 |
0.06 |
0.33 |
0.22 |
0.12 |
0.10 |
0.10 |
0.20 |
0.17 |
0.23 |
0.19 |
0.24 |
0.06 |
0.21 |
0.26 |
0.34 |
0.30 |
0.09 |
0.29 |
0.28 |
0.31 |
0.23 |
0.31 |
0.18 |
0.27 |
0.28 |
0.34 |
0.24 |
0.24 |
0.18 |
0.30 |
0.21 |
0.24 |
0.23 |
0.27 |
0.17 |
0.34 |
0.28 |
0.19 |
0.23 |
0.20 |
0.26 |
0.31 |
0.26 |
0.28 |
0.22 |
0.35 |
0.29 |
0.29 |
0.26 |
0.62 |
0.30 |
0.27 |
0.27 |
0.16 |
0.22 |
0.35 |
0.33 |
0.27 |
0.28 |
0.32 |
0.25 |
0.38 |
0.35 |
0.33 |
0.36 |
0.30 |
0.27 |
0.40 |
0.38 |
0.28 |
0.29 |
0.31 |
0.32 |
0.28 |
0.40 |
0.30 |
0.22 |
0.38 |
| Al2O3 |
0.11 |
0.15 |
0.15 |
0.24 |
0.17 |
0.22 |
0.18 |
0.36 |
0.14 |
0.12 |
0.15 |
0.10 |
0.13 |
0.17 |
0.14 |
0.10 |
0.12 |
0.15 |
0.18 |
0.15 |
0.10 |
0.11 |
0.13 |
0.16 |
0.12 |
0.16 |
0.08 |
0.11 |
0.11 |
0.10 |
0.15 |
0.16 |
0.18 |
0.12 |
0.11 |
0.02 |
0.10 |
0.12 |
0.12 |
0.07 |
0.02 |
0.09 |
0.11 |
0.05 |
0.06 |
0.09 |
0.01 |
0.07 |
0.05 |
0.05 |
0.07 |
0.08 |
0.03 |
0.07 |
0.09 |
0.08 |
0.06 |
0.03 |
0.08 |
0.05 |
0.11 |
0.07 |
0.05 |
0.09 |
0.31 |
0.05 |
0.09 |
0.05 |
0.06 |
0.04 |
0.03 |
0.07 |
0.03 |
0.08 |
0.05 |
0.08 |
0.04 |
0.02 |
0.07 |
0.08 |
0.06 |
0.07 |
0.05 |
0.05 |
0.09 |
0.06 |
0.10 |
0.10 |
0.08 |
0.05 |
0.06 |
0.08 |
0.04 |
0.04 |
| La2O3 |
0.20 |
0.08 |
0.19 |
0.08 |
0.13 |
0.15 |
0.08 |
0.17 |
0.22 |
0.16 |
0.20 |
0.14 |
0.19 |
0.15 |
0.19 |
0.18 |
0.20 |
0.21 |
0.23 |
0.09 |
0.19 |
0.11 |
0.18 |
0.10 |
0.08 |
0.12 |
0.19 |
0.18 |
0.14 |
0.22 |
0.11 |
0.10 |
0.09 |
0.13 |
0.14 |
0.74 |
0.63 |
0.39 |
0.34 |
0.58 |
0.69 |
0.65 |
0.44 |
0.70 |
0.66 |
0.67 |
0.63 |
0.67 |
0.59 |
0.75 |
0.66 |
0.76 |
0.59 |
0.63 |
0.58 |
0.62 |
0.56 |
1.14 |
0.78 |
0.66 |
0.61 |
0.82 |
1.67 |
1.40 |
1.47 |
2.29 |
1.69 |
1.63 |
1.86 |
1.94 |
1.14 |
1.47 |
1.79 |
1.58 |
1.73 |
1.25 |
2.16 |
1.98 |
1.50 |
1.45 |
1.89 |
1.71 |
1.25 |
1.55 |
1.48 |
1.29 |
1.38 |
1.31 |
1.53 |
1.85 |
1.63 |
1.22 |
1.58 |
1.66 |
| Ce2O3 |
0.46 |
0.31 |
0.37 |
0.28 |
0.36 |
0.34 |
0.28 |
0.35 |
0.39 |
0.36 |
0.42 |
0.39 |
0.33 |
0.41 |
0.40 |
0.45 |
0.40 |
0.43 |
0.43 |
0.41 |
0.43 |
0.37 |
0.41 |
0.39 |
0.32 |
0.36 |
0.38 |
0.41 |
0.52 |
0.48 |
0.33 |
0.39 |
0.40 |
0.35 |
0.38 |
1.37 |
1.04 |
0.90 |
0.87 |
1.13 |
1.62 |
1.44 |
0.81 |
1.32 |
1.37 |
1.34 |
1.35 |
1.24 |
1.24 |
1.44 |
1.30 |
1.41 |
1.31 |
1.31 |
1.19 |
1.33 |
1.34 |
2.00 |
1.43 |
1.35 |
1.28 |
1.28 |
2.76 |
2.46 |
2.56 |
3.11 |
2.80 |
2.92 |
2.95 |
3.12 |
2.41 |
2.48 |
3.05 |
2.92 |
2.77 |
2.24 |
3.03 |
2.39 |
2.79 |
2.57 |
2.84 |
2.18 |
2.09 |
2.64 |
2.55 |
2.27 |
2.45 |
2.32 |
2.57 |
3.03 |
2.72 |
2.32 |
2.76 |
2.73 |
| Pr2O3 |
— |
0.02 |
0.03 |
0.01 |
— |
0.03 |
0.03 |
0.03 |
0.02 |
0.04 |
0.03 |
0.03 |
0.03 |
0.06 |
0.03 |
0.03 |
0.01 |
— |
0.05 |
— |
0.00 |
0.00 |
0.02 |
0.07 |
0.03 |
0.01 |
0.04 |
0.04 |
0.04 |
0.06 |
0.03 |
0.04 |
0.01 |
0.02 |
0.01 |
0.07 |
0.09 |
0.08 |
0.07 |
0.06 |
0.11 |
0.08 |
0.10 |
0.10 |
0.07 |
0.12 |
0.08 |
0.08 |
0.07 |
0.06 |
0.12 |
0.13 |
0.05 |
0.07 |
0.09 |
0.04 |
0.08 |
0.14 |
0.11 |
0.10 |
0.10 |
0.09 |
0.21 |
0.18 |
0.21 |
0.25 |
0.19 |
0.28 |
0.19 |
0.24 |
0.23 |
0.18 |
0.21 |
0.21 |
0.25 |
0.18 |
0.24 |
0.20 |
0.23 |
0.19 |
0.22 |
0.20 |
0.12 |
0.19 |
0.19 |
0.21 |
0.20 |
0.19 |
0.18 |
0.25 |
0.21 |
0.15 |
0.23 |
0.21 |
| Nd2O3 |
0.13 |
0.14 |
0.10 |
0.05 |
0.08 |
0.07 |
0.08 |
0.15 |
0.14 |
0.10 |
0.18 |
0.12 |
0.07 |
0.07 |
0.13 |
0.20 |
0.10 |
0.08 |
0.08 |
0.19 |
0.10 |
0.11 |
0.09 |
0.14 |
0.10 |
0.05 |
0.11 |
0.15 |
0.17 |
0.12 |
0.15 |
0.07 |
0.22 |
0.09 |
0.09 |
0.32 |
0.21 |
0.19 |
0.23 |
0.19 |
0.30 |
0.30 |
0.18 |
0.33 |
0.32 |
0.24 |
0.33 |
0.29 |
0.34 |
0.27 |
0.32 |
0.32 |
0.24 |
0.36 |
0.23 |
0.31 |
0.35 |
0.52 |
0.29 |
0.35 |
0.32 |
0.30 |
0.49 |
0.48 |
0.40 |
0.42 |
0.49 |
0.62 |
0.51 |
0.60 |
0.62 |
0.59 |
0.60 |
0.54 |
0.51 |
0.35 |
0.51 |
0.34 |
0.71 |
0.58 |
0.45 |
0.45 |
0.33 |
0.57 |
0.56 |
0.60 |
0.44 |
0.54 |
0.49 |
0.50 |
0.51 |
0.53 |
0.47 |
0.53 |
| Sm2O3 |
0.03 |
0.03 |
0.03 |
— |
— |
— |
0.08 |
— |
0.07 |
0.03 |
0.01 |
0.05 |
0.03 |
— |
0.06 |
0.04 |
0.01 |
— |
0.05 |
0.02 |
— |
— |
0.06 |
0.02 |
0.05 |
0.05 |
0.02 |
0.01 |
0.03 |
0.04 |
— |
0.07 |
— |
— |
0.06 |
0.01 |
0.00 |
— |
0.03 |
0.00 |
0.08 |
0.02 |
0.08 |
0.04 |
0.06 |
0.07 |
0.04 |
0.04 |
0.03 |
0.08 |
0.13 |
0.04 |
0.05 |
0.12 |
— |
0.08 |
0.11 |
0.15 |
0.02 |
0.04 |
0.05 |
0.06 |
0.11 |
0.10 |
0.08 |
0.12 |
0.09 |
0.06 |
0.10 |
0.09 |
0.08 |
0.11 |
0.14 |
0.11 |
0.15 |
0.04 |
0.08 |
0.01 |
0.16 |
0.16 |
0.12 |
0.08 |
0.11 |
0.12 |
0.11 |
0.12 |
0.12 |
0.13 |
0.06 |
0.12 |
0.05 |
0.07 |
0.03 |
0.07 |
| Y2O3 |
— |
0.05 |
— |
0.03 |
0.04 |
0.06 |
0.04 |
0.13 |
0.02 |
— |
0.09 |
0.06 |
0.07 |
— |
0.08 |
0.03 |
0.04 |
0.02 |
— |
— |
0.03 |
— |
0.03 |
0.01 |
0.07 |
0.04 |
— |
0.04 |
0.10 |
0.01 |
— |
— |
0.01 |
0.08 |
— |
0.35 |
0.18 |
0.20 |
0.19 |
0.45 |
0.41 |
0.34 |
0.11 |
0.35 |
0.27 |
0.36 |
0.31 |
0.39 |
0.32 |
0.33 |
0.32 |
0.35 |
0.31 |
0.34 |
0.35 |
0.42 |
0.29 |
0.71 |
0.45 |
0.33 |
0.37 |
0.18 |
0.70 |
0.84 |
0.62 |
0.72 |
0.58 |
0.97 |
0.77 |
0.70 |
0.84 |
0.70 |
0.64 |
0.68 |
0.93 |
0.81 |
0.91 |
0.72 |
0.67 |
0.75 |
0.78 |
0.67 |
0.37 |
0.69 |
0.86 |
0.86 |
0.95 |
1.07 |
0.75 |
1.04 |
0.78 |
0.85 |
0.65 |
0.65 |
| CaO |
10.85 |
11.02 |
10.97 |
10.44 |
11.21 |
11.24 |
11.26 |
11.22 |
11.14 |
10.70 |
10.19 |
10.47 |
10.71 |
10.62 |
10.53 |
10.58 |
10.58 |
10.22 |
10.23 |
10.66 |
10.28 |
10.24 |
10.45 |
10.01 |
10.50 |
10.44 |
10.66 |
10.31 |
10.59 |
10.59 |
10.50 |
10.46 |
9.92 |
10.20 |
10.27 |
9.26 |
9.69 |
9.89 |
9.91 |
8.79 |
8.58 |
8.49 |
10.27 |
9.38 |
9.63 |
9.02 |
8.95 |
9.55 |
9.14 |
9.01 |
8.76 |
9.53 |
9.21 |
9.44 |
9.59 |
9.65 |
9.24 |
8.74 |
8.85 |
9.05 |
9.10 |
8.92 |
8.41 |
8.70 |
8.71 |
8.06 |
8.12 |
8.25 |
7.70 |
7.85 |
8.18 |
8.39 |
8.12 |
7.65 |
8.11 |
8.63 |
8.20 |
8.62 |
8.55 |
7.88 |
8.07 |
7.92 |
9.08 |
8.27 |
8.37 |
8.37 |
8.13 |
8.18 |
8.59 |
8.10 |
8.04 |
8.74 |
8.29 |
8.64 |
| MgO |
0.03 |
0.03 |
0.07 |
0.03 |
— |
0.05 |
0.02 |
0.05 |
0.01 |
0.02 |
0.05 |
0.09 |
0.06 |
0.08 |
0.05 |
0.07 |
0.06 |
0.06 |
0.05 |
0.05 |
0.07 |
0.01 |
0.07 |
0.06 |
0.06 |
0.07 |
0.07 |
0.05 |
0.05 |
0.04 |
0.04 |
0.01 |
0.06 |
0.06 |
0.03 |
0.09 |
0.08 |
0.06 |
0.08 |
0.07 |
0.10 |
0.09 |
0.05 |
0.05 |
0.07 |
0.09 |
0.09 |
0.08 |
0.08 |
0.11 |
0.08 |
0.09 |
0.08 |
0.06 |
0.09 |
0.07 |
0.11 |
0.06 |
0.11 |
0.08 |
0.06 |
0.03 |
0.08 |
0.15 |
0.08 |
0.09 |
0.07 |
0.09 |
0.07 |
0.08 |
0.04 |
0.07 |
0.10 |
0.07 |
0.06 |
0.10 |
0.06 |
0.07 |
0.08 |
0.08 |
0.06 |
0.09 |
0.06 |
0.04 |
0.08 |
0.07 |
0.10 |
0.08 |
0.04 |
0.06 |
0.06 |
0.09 |
0.08 |
0.09 |
| FeO |
6.46 |
6.74 |
6.28 |
6.37 |
6.42 |
6.73 |
6.45 |
6.64 |
6.44 |
6.50 |
6.71 |
6.11 |
6.40 |
6.44 |
6.35 |
6.16 |
6.53 |
5.92 |
6.11 |
6.25 |
6.43 |
6.61 |
6.51 |
6.60 |
6.34 |
6.80 |
6.03 |
6.45 |
6.18 |
6.29 |
6.55 |
6.61 |
6.25 |
6.26 |
6.56 |
4.59 |
6.04 |
5.48 |
5.37 |
5.60 |
5.30 |
5.23 |
5.62 |
5.36 |
5.33 |
5.27 |
5.32 |
4.97 |
4.82 |
5.56 |
5.39 |
5.04 |
4.77 |
5.11 |
5.78 |
5.07 |
4.98 |
4.74 |
5.09 |
5.32 |
5.58 |
5.38 |
3.02 |
2.63 |
2.81 |
3.50 |
3.51 |
3.00 |
3.80 |
2.72 |
2.76 |
2.80 |
3.18 |
3.26 |
3.67 |
3.71 |
4.15 |
4.04 |
3.49 |
2.73 |
3.59 |
4.40 |
2.82 |
3.23 |
3.44 |
3.02 |
3.71 |
4.09 |
3.57 |
3.50 |
3.19 |
3.13 |
3.34 |
3.33 |
| MnO |
1.38 |
1.26 |
1.26 |
1.14 |
1.21 |
1.20 |
1.18 |
1.41 |
1.29 |
1.23 |
0.96 |
1.22 |
1.19 |
1.10 |
1.28 |
1.28 |
1.20 |
1.07 |
1.05 |
1.27 |
1.29 |
1.26 |
1.30 |
1.24 |
1.25 |
1.16 |
1.36 |
1.18 |
1.28 |
1.33 |
1.20 |
1.10 |
1.04 |
1.20 |
1.19 |
2.10 |
2.08 |
1.69 |
1.82 |
2.04 |
2.19 |
2.00 |
1.56 |
2.22 |
2.28 |
2.37 |
2.10 |
2.02 |
1.97 |
2.00 |
2.12 |
2.01 |
2.08 |
2.14 |
1.99 |
1.93 |
1.88 |
2.04 |
2.17 |
2.15 |
1.99 |
2.17 |
3.51 |
3.64 |
3.63 |
3.88 |
3.34 |
3.79 |
3.60 |
4.51 |
4.39 |
3.63 |
3.90 |
3.94 |
3.40 |
3.04 |
2.98 |
2.86 |
3.35 |
3.81 |
3.07 |
2.71 |
4.34 |
3.93 |
3.17 |
3.79 |
2.93 |
2.99 |
3.47 |
3.44 |
3.72 |
3.68 |
3.74 |
3.19 |
| SrO |
2.35 |
1.96 |
2.47 |
1.91 |
1.92 |
1.82 |
1.93 |
1.80 |
2.32 |
2.24 |
2.43 |
2.37 |
2.38 |
2.35 |
2.34 |
2.11 |
2.29 |
2.19 |
1.95 |
2.27 |
2.25 |
2.19 |
2.42 |
2.31 |
2.26 |
2.07 |
2.35 |
2.27 |
2.11 |
2.08 |
1.92 |
2.08 |
2.09 |
2.04 |
2.42 |
3.55 |
2.96 |
3.22 |
3.03 |
3.33 |
4.38 |
3.74 |
3.08 |
3.36 |
3.38 |
3.23 |
3.65 |
3.66 |
4.00 |
3.88 |
3.47 |
3.08 |
3.54 |
3.82 |
3.41 |
3.70 |
3.64 |
3.53 |
3.57 |
3.37 |
3.48 |
3.83 |
2.49 |
2.63 |
2.99 |
2.49 |
2.97 |
3.00 |
2.89 |
3.14 |
3.55 |
3.05 |
2.70 |
2.70 |
2.49 |
2.90 |
2.54 |
2.89 |
2.58 |
3.42 |
2.58 |
2.74 |
3.50 |
2.91 |
2.63 |
3.06 |
2.79 |
2.60 |
2.82 |
2.23 |
2.77 |
2.86 |
2.73 |
2.43 |
| Na2O |
9.27 |
8.41 |
8.24 |
7.30 |
9.69 |
9.03 |
9.67 |
8.79 |
10.74 |
10.37 |
9.38 |
12.62 |
9.77 |
10.14 |
10.22 |
10.57 |
9.82 |
14.03 |
12.31 |
11.00 |
10.55 |
9.76 |
9.95 |
9.92 |
13.68 |
10.69 |
11.75 |
10.90 |
12.36 |
10.22 |
10.54 |
10.20 |
13.66 |
13.85 |
10.91 |
10.86 |
4.62 |
11.69 |
11.48 |
3.33 |
7.66 |
7.54 |
8.81 |
10.84 |
7.45 |
10.65 |
10.76 |
11.25 |
10.69 |
10.80 |
11.54 |
10.09 |
9.43 |
11.09 |
11.19 |
11.62 |
11.12 |
11.09 |
6.63 |
5.30 |
3.28 |
4.84 |
11.20 |
11.02 |
10.04 |
4.73 |
4.58 |
8.71 |
4.09 |
5.55 |
4.35 |
7.47 |
5.72 |
8.06 |
4.88 |
2.94 |
4.81 |
5.01 |
8.18 |
7.40 |
6.32 |
3.60 |
4.25 |
9.77 |
4.75 |
4.87 |
5.68 |
8.55 |
4.91 |
6.33 |
3.99 |
5.51 |
5.01 |
10.90 |
| K2O |
0.22 |
0.22 |
0.16 |
0.22 |
0.23 |
0.21 |
0.21 |
0.22 |
0.22 |
0.45 |
0.25 |
0.22 |
0.21 |
0.34 |
0.33 |
0.29 |
0.29 |
0.41 |
0.25 |
0.21 |
0.26 |
0.25 |
0.22 |
0.18 |
0.32 |
0.30 |
0.20 |
0.27 |
0.24 |
0.26 |
0.39 |
0.34 |
0.52 |
0.46 |
0.33 |
0.41 |
0.31 |
0.34 |
0.35 |
0.23 |
0.23 |
0.23 |
0.31 |
0.35 |
0.28 |
0.33 |
0.32 |
0.36 |
0.38 |
0.35 |
0.31 |
0.45 |
0.35 |
0.38 |
0.35 |
0.36 |
0.35 |
0.39 |
0.25 |
0.23 |
0.24 |
0.24 |
0.74 |
0.68 |
1.21 |
0.41 |
0.60 |
0.56 |
0.44 |
0.45 |
0.45 |
0.51 |
0.55 |
0.53 |
0.55 |
0.51 |
0.72 |
0.59 |
0.56 |
0.49 |
0.50 |
0.67 |
0.57 |
0.41 |
0.57 |
0.46 |
0.62 |
0.69 |
0.59 |
0.41 |
0.46 |
0.46 |
0.41 |
0.79 |
| F |
— |
0.01 |
0.14 |
0.13 |
— |
0.09 |
0.08 |
— |
— |
0.02 |
0.01 |
— |
— |
— |
0.01 |
0.20 |
0.27 |
0.27 |
0.23 |
— |
— |
0.03 |
— |
— |
— |
— |
— |
0.01 |
— |
0.02 |
— |
— |
0.01 |
— |
— |
0.12 |
0.23 |
— |
0.01 |
0.24 |
0.04 |
0.13 |
— |
— |
0.03 |
0.03 |
0.02 |
— |
0.03 |
0.23 |
0.21 |
— |
0.23 |
0.17 |
— |
— |
— |
0.03 |
0.01 |
0.03 |
— |
0.19 |
— |
0.12 |
0.04 |
0.05 |
0.02 |
— |
0.19 |
— |
— |
0.24 |
0.15 |
— |
0.26 |
0.04 |
0.08 |
0.02 |
— |
0.02 |
0.04 |
0.29 |
0.19 |
— |
0.24 |
0.28 |
— |
— |
0.31 |
0.05 |
0.08 |
0.07 |
— |
0.01 |
| Cl |
1.21 |
1.16 |
1.25 |
1.30 |
1.25 |
1.17 |
1.13 |
1.11 |
1.24 |
1.33 |
1.31 |
1.38 |
1.35 |
1.34 |
1.30 |
1.36 |
1.41 |
1.34 |
1.26 |
1.32 |
1.24 |
1.29 |
1.20 |
1.25 |
1.21 |
1.20 |
1.18 |
1.30 |
1.27 |
1.15 |
1.20 |
1.24 |
1.33 |
1.18 |
1.25 |
1.18 |
1.26 |
1.16 |
1.17 |
1.26 |
1.14 |
1.24 |
1.26 |
1.16 |
1.16 |
1.23 |
1.25 |
1.19 |
1.19 |
1.13 |
1.22 |
0.81 |
1.21 |
1.22 |
1.21 |
1.19 |
1.10 |
0.93 |
1.21 |
1.19 |
0.00 |
1.28 |
0.71 |
0.82 |
0.69 |
0.60 |
0.69 |
0.72 |
0.66 |
0.74 |
0.76 |
0.82 |
0.76 |
0.85 |
0.76 |
0.78 |
0.47 |
0.55 |
0.67 |
0.73 |
0.69 |
0.65 |
0.74 |
0.85 |
0.72 |
0.73 |
0.74 |
0.80 |
0.68 |
0.74 |
0.83 |
0.79 |
0.75 |
0.62 |
| F=O |
— |
0.01 |
0.06 |
0.06 |
— |
0.04 |
0.03 |
— |
— |
0.01 |
0.01 |
— |
— |
— |
— |
0.08 |
0.11 |
0.11 |
0.10 |
— |
— |
0.01 |
— |
— |
— |
— |
— |
0.01 |
— |
0.01 |
— |
— |
0.01 |
— |
— |
0.05 |
0.10 |
— |
0.01 |
0.10 |
0.02 |
0.05 |
— |
— |
0.01 |
0.01 |
0.01 |
— |
0.01 |
0.10 |
0.09 |
— |
0.10 |
0.07 |
— |
— |
— |
0.01 |
0.01 |
0.01 |
— |
0.08 |
— |
0.05 |
0.02 |
0.02 |
0.01 |
— |
0.08 |
— |
— |
0.10 |
0.06 |
— |
0.11 |
0.01 |
0.03 |
0.01 |
— |
0.01 |
0.02 |
0.12 |
0.08 |
— |
0.10 |
0.12 |
— |
— |
0.13 |
0.02 |
0.03 |
0.03 |
— |
— |
| Cl=O |
0.28 |
0.27 |
0.29 |
0.30 |
0.29 |
0.27 |
0.26 |
0.26 |
0.28 |
0.31 |
0.30 |
0.32 |
0.31 |
0.31 |
0.30 |
0.31 |
0.32 |
0.31 |
0.29 |
0.30 |
0.29 |
0.30 |
0.28 |
0.29 |
0.28 |
0.28 |
0.27 |
0.30 |
0.29 |
0.26 |
0.28 |
0.28 |
0.30 |
0.27 |
0.29 |
0.27 |
0.29 |
0.27 |
0.27 |
0.29 |
0.26 |
0.28 |
0.29 |
0.27 |
0.27 |
0.28 |
0.29 |
0.27 |
0.27 |
0.26 |
0.28 |
0.19 |
0.28 |
0.28 |
0.28 |
0.27 |
0.25 |
0.21 |
0.28 |
0.27 |
0.29 |
0.30 |
0.16 |
0.19 |
0.16 |
0.14 |
0.16 |
0.17 |
0.15 |
0.17 |
0.17 |
0.19 |
0.18 |
0.20 |
0.18 |
0.18 |
0.11 |
0.13 |
0.15 |
0.17 |
0.16 |
0.15 |
0.17 |
0.20 |
0.17 |
0.17 |
0.17 |
0.18 |
0.16 |
0.17 |
0.19 |
0.18 |
0.17 |
0.14 |
| Total |
99.83 |
99.17 |
99.03 |
98.27 |
100.68 |
99.94 |
100.98 |
99.22 |
101.80 |
100.86 |
99.60 |
97.26 |
100.22 |
100.35 |
100.45 |
99.82 |
99.86 |
98.69 |
98.06 |
101.16 |
99.49 |
99.01 |
100.17 |
99.88 |
101.91 |
100.84 |
100.66 |
100.36 |
101.46 |
99.07 |
99.77 |
100.39 |
99.78 |
99.78 |
100.34 |
95.22 |
95.11 |
97.62 |
97.94 |
91.90 |
96.48 |
94.32 |
97.68 |
98.18 |
94.76 |
96.60 |
96.86 |
97.61 |
95.41 |
96.33 |
96.64 |
98.04 |
94.05 |
96.32 |
99.86 |
99.15 |
96.60 |
98.04 |
95.82 |
95.84 |
92.85 |
96.38 |
97.12 |
97.21 |
97.76 |
97.52 |
95.68 |
98.00 |
96.02 |
95.59 |
95.12 |
96.59 |
96.44 |
97.18 |
96.02 |
94.23 |
96.64 |
96.24 |
99.34 |
97.89 |
96.34 |
94.32 |
95.13 |
97.60 |
96.20 |
95.52 |
96.08 |
98.34 |
95.50 |
97.82 |
94.87 |
96.07 |
97.22 |
96.52 |
| 根据 ∑(Nb+Si+Ti+Zr+Hf+Al) =29 a.p.f.u计算 |
| Nb |
0.211 |
0.094 |
0.133 |
0.075 |
0.057 |
0.045 |
0.097 |
0.056 |
0.137 |
0.176 |
0.156 |
0.169 |
0.150 |
0.181 |
0.205 |
0.158 |
0.194 |
0.175 |
0.141 |
0.130 |
0.175 |
0.148 |
0.149 |
0.127 |
0.158 |
0.126 |
0.198 |
0.158 |
0.160 |
0.178 |
0.103 |
0.099 |
0.150 |
0.122 |
0.147 |
0.808 |
0.576 |
0.515 |
0.511 |
0.551 |
0.905 |
0.765 |
0.363 |
0.717 |
0.687 |
0.756 |
0.772 |
0.794 |
0.902 |
0.834 |
0.730 |
0.665 |
0.786 |
0.810 |
0.630 |
0.725 |
0.719 |
0.826 |
0.790 |
0.652 |
0.623 |
0.754 |
0.771 |
0.623 |
0.802 |
0.909 |
0.772 |
0.792 |
0.813 |
0.853 |
0.614 |
0.657 |
0.838 |
0.704 |
0.769 |
0.681 |
0.822 |
0.856 |
0.747 |
0.716 |
0.770 |
0.740 |
0.756 |
0.659 |
0.760 |
0.620 |
0.660 |
0.568 |
0.689 |
0.757 |
0.810 |
0.570 |
0.754 |
0.771 |
| Ta |
0.005 |
0.029 |
— |
0.016 |
0.010 |
0.027 |
0.001 |
— |
0.024 |
0.003 |
— |
0.004 |
0.016 |
0.023 |
0.009 |
— |
— |
0.035 |
0.005 |
0.019 |
0.017 |
— |
0.006 |
— |
0.022 |
— |
0.013 |
— |
0.024 |
0.005 |
— |
— |
— |
— |
0.002 |
0.039 |
0.032 |
0.022 |
— |
0.007 |
0.018 |
— |
0.012 |
0.022 |
0.036 |
0.017 |
0.032 |
— |
— |
0.004 |
0.002 |
— |
0.041 |
— |
0.002 |
0.023 |
0.010 |
— |
0.006 |
0.039 |
0.005 |
0.048 |
0.032 |
0.035 |
0.022 |
— |
0.034 |
0.005 |
0.002 |
— |
0.026 |
— |
— |
0.002 |
0.032 |
0.029 |
0.014 |
0.017 |
— |
— |
— |
0.017 |
0.039 |
— |
0.020 |
— |
— |
— |
0.032 |
0.008 |
0.029 |
0.032 |
— |
0.005 |
| Si |
25.734 |
25.820 |
25.667 |
25.610 |
25.744 |
25.789 |
25.700 |
25.588 |
25.730 |
25.702 |
25.660 |
25.749 |
25.737 |
25.692 |
25.617 |
25.825 |
25.719 |
25.592 |
25.552 |
25.677 |
25.671 |
25.663 |
25.580 |
25.667 |
25.656 |
25.665 |
25.672 |
25.696 |
25.570 |
25.655 |
25.787 |
25.694 |
25.563 |
25.645 |
25.712 |
25.046 |
25.245 |
25.286 |
25.264 |
25.252 |
25.091 |
25.022 |
25.393 |
25.003 |
25.183 |
25.050 |
25.039 |
24.999 |
24.972 |
25.045 |
25.144 |
25.114 |
25.039 |
25.276 |
25.213 |
25.142 |
25.061 |
24.897 |
25.074 |
25.138 |
25.170 |
24.961 |
24.879 |
24.930 |
24.715 |
24.869 |
24.907 |
24.940 |
24.934 |
24.912 |
25.045 |
24.902 |
24.889 |
24.887 |
25.037 |
25.042 |
25.120 |
25.047 |
24.775 |
24.876 |
24.945 |
25.119 |
24.953 |
25.082 |
24.823 |
24.985 |
25.074 |
25.077 |
25.098 |
24.895 |
24.812 |
24.912 |
24.835 |
24.993 |
| Ti |
0.155 |
0.321 |
0.120 |
0.180 |
0.176 |
0.288 |
0.214 |
0.271 |
0.212 |
0.160 |
0.136 |
0.114 |
0.139 |
0.137 |
0.143 |
0.136 |
0.121 |
0.173 |
0.127 |
0.121 |
0.141 |
0.144 |
0.159 |
0.146 |
0.134 |
0.173 |
0.159 |
0.161 |
0.145 |
0.142 |
0.153 |
0.166 |
0.131 |
0.144 |
0.132 |
0.039 |
0.095 |
0.115 |
0.087 |
0.097 |
0.043 |
0.046 |
0.137 |
0.078 |
0.049 |
0.059 |
0.041 |
0.052 |
0.041 |
0.046 |
0.080 |
0.097 |
0.057 |
0.041 |
0.090 |
0.053 |
0.078 |
0.065 |
0.048 |
0.082 |
0.099 |
0.068 |
0.165 |
0.274 |
0.137 |
0.105 |
0.150 |
0.225 |
0.161 |
0.165 |
0.357 |
0.290 |
0.172 |
0.240 |
0.137 |
0.180 |
0.061 |
0.060 |
0.240 |
0.263 |
0.147 |
0.070 |
0.256 |
0.211 |
0.188 |
0.288 |
0.175 |
0.244 |
0.189 |
0.166 |
0.164 |
0.358 |
0.186 |
0.132 |
| Zr |
2.815 |
2.667 |
2.977 |
2.975 |
2.905 |
2.732 |
2.860 |
2.860 |
2.814 |
2.874 |
2.947 |
2.866 |
2.870 |
2.885 |
2.909 |
2.797 |
2.892 |
2.922 |
3.043 |
2.972 |
2.943 |
2.968 |
3.010 |
2.948 |
2.949 |
2.919 |
2.893 |
2.917 |
3.030 |
2.931 |
2.826 |
2.913 |
3.036 |
2.972 |
2.908 |
3.042 |
2.990 |
2.964 |
3.036 |
3.018 |
2.904 |
3.060 |
3.008 |
3.135 |
3.014 |
3.034 |
3.110 |
3.074 |
3.014 |
2.999 |
2.972 |
3.025 |
3.056 |
2.797 |
2.977 |
3.003 |
3.067 |
3.149 |
3.005 |
3.058 |
3.011 |
3.125 |
3.111 |
3.072 |
3.110 |
3.001 |
3.071 |
2.974 |
3.020 |
3.021 |
2.937 |
3.059 |
3.035 |
3.077 |
2.988 |
3.004 |
2.936 |
2.970 |
3.148 |
3.052 |
3.051 |
2.989 |
2.964 |
2.956 |
3.119 |
3.029 |
2.991 |
3.005 |
2.932 |
3.113 |
3.122 |
3.071 |
3.173 |
3.020 |
| Hf |
0.025 |
0.012 |
0.018 |
0.028 |
0.025 |
0.024 |
0.027 |
0.023 |
0.030 |
0.019 |
0.016 |
0.040 |
0.031 |
0.007 |
0.049 |
0.029 |
0.008 |
0.049 |
0.031 |
0.017 |
0.015 |
0.014 |
0.028 |
0.024 |
0.033 |
0.025 |
0.033 |
0.008 |
0.030 |
0.036 |
0.047 |
0.041 |
0.013 |
0.042 |
0.038 |
0.049 |
0.033 |
0.046 |
0.026 |
0.039 |
0.041 |
0.050 |
0.034 |
0.036 |
0.027 |
0.047 |
0.031 |
0.036 |
0.037 |
0.042 |
0.027 |
0.050 |
0.045 |
0.030 |
0.033 |
0.030 |
0.040 |
0.047 |
0.037 |
0.040 |
0.032 |
0.049 |
0.045 |
0.045 |
0.040 |
0.089 |
0.043 |
0.040 |
0.038 |
0.023 |
0.031 |
0.051 |
0.048 |
0.040 |
0.040 |
0.046 |
0.036 |
0.054 |
0.050 |
0.048 |
0.053 |
0.044 |
0.039 |
0.061 |
0.055 |
0.041 |
0.041 |
0.045 |
0.047 |
0.039 |
0.058 |
0.043 |
0.030 |
0.058 |
| Al |
0.061 |
0.085 |
0.085 |
0.132 |
0.094 |
0.122 |
0.101 |
0.201 |
0.077 |
0.068 |
0.085 |
0.061 |
0.073 |
0.098 |
0.077 |
0.055 |
0.066 |
0.090 |
0.106 |
0.084 |
0.055 |
0.063 |
0.075 |
0.089 |
0.070 |
0.092 |
0.046 |
0.059 |
0.065 |
0.059 |
0.084 |
0.087 |
0.107 |
0.074 |
0.062 |
0.015 |
0.060 |
0.074 |
0.075 |
0.044 |
0.015 |
0.057 |
0.065 |
0.031 |
0.039 |
0.055 |
0.007 |
0.045 |
0.035 |
0.033 |
0.047 |
0.048 |
0.018 |
0.046 |
0.057 |
0.047 |
0.036 |
0.016 |
0.045 |
0.030 |
0.066 |
0.043 |
0.030 |
0.055 |
0.196 |
0.029 |
0.056 |
0.028 |
0.033 |
0.026 |
0.016 |
0.041 |
0.019 |
0.052 |
0.030 |
0.048 |
0.025 |
0.012 |
0.039 |
0.045 |
0.034 |
0.039 |
0.032 |
0.031 |
0.054 |
0.036 |
0.058 |
0.061 |
0.045 |
0.030 |
0.033 |
0.046 |
0.021 |
0.026 |
| La |
0.035 |
0.014 |
0.034 |
0.014 |
0.023 |
0.025 |
0.013 |
0.030 |
0.039 |
0.028 |
0.036 |
0.027 |
0.034 |
0.027 |
0.034 |
0.031 |
0.035 |
0.039 |
0.042 |
0.015 |
0.035 |
0.020 |
0.032 |
0.017 |
0.014 |
0.020 |
0.035 |
0.032 |
0.024 |
0.040 |
0.019 |
0.017 |
0.016 |
0.023 |
0.024 |
0.151 |
0.116 |
0.076 |
0.066 |
0.108 |
0.133 |
0.126 |
0.082 |
0.137 |
0.129 |
0.134 |
0.125 |
0.132 |
0.118 |
0.151 |
0.131 |
0.144 |
0.120 |
0.127 |
0.110 |
0.120 |
0.111 |
0.227 |
0.146 |
0.121 |
0.110 |
0.151 |
0.337 |
0.280 |
0.291 |
0.423 |
0.316 |
0.314 |
0.344 |
0.373 |
0.212 |
0.279 |
0.338 |
0.302 |
0.324 |
0.230 |
0.402 |
0.369 |
0.280 |
0.269 |
0.355 |
0.316 |
0.235 |
0.301 |
0.273 |
0.240 |
0.256 |
0.246 |
0.287 |
0.342 |
0.307 |
0.227 |
0.289 |
0.334 |
| Ce |
0.080 |
0.053 |
0.066 |
0.048 |
0.061 |
0.059 |
0.048 |
0.061 |
0.069 |
0.064 |
0.074 |
0.074 |
0.058 |
0.072 |
0.070 |
0.079 |
0.071 |
0.080 |
0.080 |
0.071 |
0.077 |
0.065 |
0.073 |
0.068 |
0.058 |
0.063 |
0.068 |
0.072 |
0.092 |
0.086 |
0.059 |
0.068 |
0.074 |
0.066 |
0.067 |
0.278 |
0.191 |
0.173 |
0.167 |
0.211 |
0.308 |
0.278 |
0.149 |
0.257 |
0.265 |
0.263 |
0.267 |
0.244 |
0.248 |
0.289 |
0.259 |
0.267 |
0.264 |
0.262 |
0.225 |
0.256 |
0.265 |
0.395 |
0.267 |
0.246 |
0.233 |
0.234 |
0.552 |
0.488 |
0.502 |
0.568 |
0.518 |
0.559 |
0.542 |
0.594 |
0.446 |
0.468 |
0.572 |
0.553 |
0.514 |
0.407 |
0.559 |
0.441 |
0.518 |
0.475 |
0.530 |
0.400 |
0.390 |
0.510 |
0.468 |
0.419 |
0.449 |
0.430 |
0.479 |
0.558 |
0.507 |
0.428 |
0.500 |
0.548 |
| Pr |
— |
0.004 |
0.005 |
0.001 |
— |
0.005 |
0.006 |
0.005 |
0.003 |
0.007 |
0.005 |
0.005 |
0.005 |
0.011 |
0.006 |
0.005 |
0.003 |
— |
0.009 |
— |
0.001 |
0.001 |
0.003 |
0.011 |
0.005 |
0.002 |
0.006 |
0.007 |
0.006 |
0.010 |
0.006 |
0.007 |
0.002 |
0.004 |
0.002 |
0.013 |
0.017 |
0.016 |
0.013 |
0.012 |
0.022 |
0.015 |
0.019 |
0.020 |
0.012 |
0.023 |
0.015 |
0.015 |
0.013 |
0.012 |
0.023 |
0.025 |
0.010 |
0.014 |
0.016 |
0.007 |
0.016 |
0.027 |
0.019 |
0.018 |
0.017 |
0.016 |
0.041 |
0.037 |
0.040 |
0.045 |
0.034 |
0.054 |
0.035 |
0.047 |
0.041 |
0.034 |
0.038 |
0.039 |
0.047 |
0.033 |
0.043 |
0.036 |
0.042 |
0.035 |
0.041 |
0.035 |
0.022 |
0.037 |
0.035 |
0.038 |
0.036 |
0.036 |
0.034 |
0.047 |
0.038 |
0.027 |
0.041 |
0.042 |
| Nd |
0.022 |
0.023 |
0.017 |
0.009 |
0.014 |
0.012 |
0.012 |
0.024 |
0.023 |
0.016 |
0.031 |
0.022 |
0.011 |
0.012 |
0.022 |
0.034 |
0.016 |
0.014 |
0.015 |
0.032 |
0.017 |
0.019 |
0.015 |
0.025 |
0.019 |
0.008 |
0.018 |
0.025 |
0.029 |
0.021 |
0.026 |
0.012 |
0.039 |
0.017 |
0.016 |
0.063 |
0.038 |
0.036 |
0.044 |
0.034 |
0.055 |
0.056 |
0.033 |
0.062 |
0.062 |
0.046 |
0.064 |
0.055 |
0.067 |
0.052 |
0.063 |
0.058 |
0.046 |
0.070 |
0.042 |
0.058 |
0.066 |
0.101 |
0.053 |
0.063 |
0.057 |
0.054 |
0.096 |
0.092 |
0.076 |
0.075 |
0.088 |
0.116 |
0.091 |
0.111 |
0.112 |
0.108 |
0.109 |
0.100 |
0.092 |
0.062 |
0.092 |
0.061 |
0.129 |
0.103 |
0.083 |
0.080 |
0.060 |
0.108 |
0.099 |
0.107 |
0.078 |
0.098 |
0.089 |
0.089 |
0.093 |
0.096 |
0.082 |
0.104 |
| Sm |
0.005 |
0.006 |
0.006 |
— |
— |
— |
0.013 |
— |
0.013 |
0.005 |
0.002 |
0.009 |
0.005 |
— |
0.010 |
0.008 |
0.001 |
— |
0.008 |
0.003 |
— |
— |
0.010 |
0.003 |
0.008 |
0.008 |
0.004 |
0.001 |
0.004 |
0.007 |
— |
0.011 |
— |
— |
0.010 |
0.002 |
0.001 |
— |
0.005 |
0.001 |
0.014 |
0.004 |
0.014 |
0.008 |
0.011 |
0.014 |
0.007 |
0.007 |
0.005 |
0.015 |
0.024 |
0.008 |
0.010 |
0.022 |
— |
0.013 |
0.019 |
0.029 |
0.005 |
0.007 |
0.008 |
0.010 |
0.021 |
0.019 |
0.014 |
0.021 |
0.016 |
0.012 |
0.017 |
0.015 |
0.014 |
0.019 |
0.024 |
0.020 |
0.026 |
0.006 |
0.014 |
0.003 |
0.028 |
0.028 |
0.022 |
0.014 |
0.018 |
0.021 |
0.020 |
0.022 |
0.020 |
0.023 |
0.010 |
0.021 |
0.009 |
0.012 |
0.006 |
0.013 |
| Y |
— |
0.012 |
— |
0.008 |
0.010 |
0.014 |
0.009 |
0.032 |
0.005 |
— |
0.024 |
0.017 |
0.018 |
— |
0.021 |
0.009 |
0.011 |
0.005 |
— |
— |
0.009 |
— |
0.007 |
0.002 |
0.019 |
0.010 |
— |
0.010 |
0.025 |
0.002 |
— |
— |
0.002 |
0.022 |
— |
0.104 |
0.048 |
0.056 |
0.054 |
0.121 |
0.113 |
0.096 |
0.029 |
0.097 |
0.076 |
0.104 |
0.089 |
0.110 |
0.093 |
0.097 |
0.092 |
0.097 |
0.091 |
0.100 |
0.096 |
0.118 |
0.084 |
0.203 |
0.123 |
0.089 |
0.097 |
0.048 |
0.204 |
0.242 |
0.178 |
0.193 |
0.156 |
0.270 |
0.206 |
0.195 |
0.227 |
0.194 |
0.176 |
0.187 |
0.252 |
0.215 |
0.243 |
0.194 |
0.179 |
0.201 |
0.211 |
0.178 |
0.099 |
0.193 |
0.229 |
0.230 |
0.254 |
0.289 |
0.202 |
0.278 |
0.211 |
0.227 |
0.173 |
0.189 |
| Ca |
5.568 |
5.582 |
5.608 |
5.224 |
5.662 |
5.688 |
5.656 |
5.739 |
5.691 |
5.481 |
5.217 |
5.827 |
5.482 |
5.452 |
5.434 |
5.482 |
5.460 |
5.676 |
5.549 |
5.470 |
5.360 |
5.296 |
5.385 |
5.107 |
5.540 |
5.334 |
5.566 |
5.320 |
5.542 |
5.540 |
5.410 |
5.330 |
5.347 |
5.521 |
5.316 |
5.484 |
5.195 |
5.584 |
5.522 |
4.777 |
4.794 |
4.793 |
5.524 |
5.329 |
5.465 |
5.199 |
5.167 |
5.486 |
5.373 |
5.295 |
5.090 |
5.280 |
5.410 |
5.527 |
5.287 |
5.446 |
5.339 |
5.048 |
4.835 |
4.848 |
4.828 |
4.770 |
4.929 |
5.037 |
4.998 |
4.319 |
4.402 |
4.627 |
4.135 |
4.375 |
4.430 |
4.635 |
4.461 |
4.241 |
4.404 |
4.596 |
4.435 |
4.665 |
4.636 |
4.268 |
4.409 |
4.255 |
4.946 |
4.687 |
4.496 |
4.516 |
4.367 |
4.452 |
4.683 |
4.365 |
4.379 |
4.731 |
4.402 |
5.073 |
| Mg |
0.019 |
0.023 |
0.046 |
0.018 |
— |
0.033 |
0.017 |
0.034 |
0.007 |
0.017 |
0.039 |
0.066 |
0.044 |
0.057 |
0.033 |
0.049 |
0.045 |
0.046 |
0.039 |
0.035 |
0.050 |
0.010 |
0.048 |
0.040 |
0.041 |
0.046 |
0.053 |
0.037 |
0.035 |
0.030 |
0.028 |
0.008 |
0.043 |
0.048 |
0.018 |
0.073 |
0.058 |
0.047 |
0.065 |
0.050 |
0.075 |
0.070 |
0.034 |
0.041 |
0.055 |
0.072 |
0.070 |
0.064 |
0.065 |
0.092 |
0.068 |
0.066 |
0.067 |
0.051 |
0.065 |
0.054 |
0.086 |
0.050 |
0.084 |
0.059 |
0.046 |
0.023 |
0.061 |
0.117 |
0.063 |
0.066 |
0.051 |
0.071 |
0.055 |
0.062 |
0.033 |
0.052 |
0.079 |
0.055 |
0.043 |
0.073 |
0.048 |
0.049 |
0.063 |
0.058 |
0.043 |
0.070 |
0.044 |
0.034 |
0.060 |
0.050 |
0.073 |
0.058 |
0.032 |
0.041 |
0.042 |
0.064 |
0.056 |
0.073 |
| Fe |
2.589 |
2.665 |
2.505 |
2.490 |
2.532 |
2.658 |
2.528 |
2.653 |
2.570 |
2.598 |
2.681 |
2.652 |
2.556 |
2.580 |
2.557 |
2.491 |
2.629 |
2.564 |
2.588 |
2.503 |
2.615 |
2.670 |
2.619 |
2.628 |
2.612 |
2.710 |
2.456 |
2.596 |
2.526 |
2.568 |
2.633 |
2.628 |
2.630 |
2.644 |
2.652 |
2.123 |
2.527 |
2.414 |
2.337 |
2.377 |
2.310 |
2.307 |
2.357 |
2.376 |
2.363 |
2.371 |
2.400 |
2.231 |
2.210 |
2.550 |
2.444 |
2.180 |
2.187 |
2.335 |
2.487 |
2.234 |
2.245 |
2.138 |
2.171 |
2.222 |
2.310 |
2.246 |
1.381 |
1.190 |
1.261 |
1.465 |
1.486 |
1.311 |
1.593 |
1.182 |
1.166 |
1.207 |
1.363 |
1.408 |
1.554 |
1.540 |
1.750 |
1.706 |
1.478 |
1.152 |
1.533 |
1.844 |
1.201 |
1.429 |
1.441 |
1.271 |
1.556 |
1.738 |
1.519 |
1.470 |
1.358 |
1.323 |
1.385 |
1.527 |
| Mn |
0.562 |
0.504 |
0.509 |
0.451 |
0.483 |
0.482 |
0.466 |
0.569 |
0.520 |
0.497 |
0.387 |
0.537 |
0.482 |
0.448 |
0.523 |
0.524 |
0.488 |
0.469 |
0.449 |
0.515 |
0.531 |
0.516 |
0.532 |
0.499 |
0.520 |
0.469 |
0.560 |
0.480 |
0.530 |
0.551 |
0.489 |
0.441 |
0.443 |
0.515 |
0.488 |
0.983 |
0.882 |
0.752 |
0.799 |
0.875 |
0.967 |
0.892 |
0.663 |
0.998 |
1.022 |
1.081 |
0.957 |
0.919 |
0.913 |
0.930 |
0.973 |
0.882 |
0.966 |
0.989 |
0.865 |
0.863 |
0.860 |
0.932 |
0.938 |
0.912 |
0.837 |
0.916 |
1.628 |
1.665 |
1.647 |
1.645 |
1.432 |
1.682 |
1.530 |
1.989 |
1.877 |
1.587 |
1.692 |
1.725 |
1.462 |
1.278 |
1.276 |
1.225 |
1.436 |
1.630 |
1.327 |
1.150 |
1.871 |
1.759 |
1.344 |
1.614 |
1.245 |
1.289 |
1.497 |
1.464 |
1.599 |
1.573 |
1.568 |
1.482 |
| Sr |
0.653 |
0.537 |
0.683 |
0.517 |
0.523 |
0.497 |
0.525 |
0.497 |
0.641 |
0.622 |
0.675 |
0.712 |
0.658 |
0.654 |
0.654 |
0.590 |
0.640 |
0.658 |
0.571 |
0.631 |
0.636 |
0.612 |
0.675 |
0.638 |
0.645 |
0.572 |
0.662 |
0.633 |
0.597 |
0.588 |
0.536 |
0.574 |
0.609 |
0.597 |
0.679 |
1.139 |
0.860 |
0.984 |
0.912 |
0.979 |
1.325 |
1.142 |
0.895 |
1.034 |
1.040 |
1.007 |
1.142 |
1.138 |
1.271 |
1.235 |
1.090 |
0.923 |
1.125 |
1.211 |
1.018 |
1.129 |
1.138 |
1.102 |
1.055 |
0.977 |
1.000 |
1.109 |
0.789 |
0.824 |
0.931 |
0.724 |
0.869 |
0.910 |
0.839 |
0.948 |
1.040 |
0.911 |
0.804 |
0.811 |
0.731 |
0.834 |
0.742 |
0.845 |
0.756 |
1.001 |
0.762 |
0.795 |
1.033 |
0.891 |
0.766 |
0.892 |
0.811 |
0.767 |
0.834 |
0.651 |
0.815 |
0.838 |
0.785 |
0.771 |
| Na |
8.609 |
7.705 |
7.624 |
6.612 |
8.862 |
8.270 |
8.794 |
8.138 |
9.926 |
9.612 |
8.694 |
12.704 |
9.048 |
9.421 |
9.547 |
9.910 |
9.176 |
14.093 |
12.080 |
10.213 |
9.957 |
9.137 |
9.273 |
9.159 |
13.063 |
9.881 |
11.098 |
10.178 |
11.703 |
9.670 |
9.824 |
9.400 |
13.316 |
13.566 |
10.222 |
11.635 |
4.479 |
11.933 |
11.576 |
3.274 |
7.742 |
7.707 |
8.576 |
11.147 |
7.651 |
11.113 |
11.246 |
11.699 |
11.365 |
11.484 |
12.129 |
10.111 |
10.028 |
11.752 |
11.171 |
11.865 |
11.625 |
11.594 |
6.561 |
5.132 |
3.149 |
4.688 |
11.884 |
11.555 |
10.427 |
4.583 |
4.490 |
8.835 |
3.975 |
5.598 |
4.266 |
7.472 |
5.689 |
8.085 |
4.796 |
2.836 |
4.708 |
4.903 |
8.026 |
7.254 |
6.253 |
3.498 |
4.189 |
10.012 |
4.616 |
4.754 |
5.521 |
8.429 |
4.849 |
6.165 |
3.930 |
5.396 |
4.816 |
11.578 |
| K |
0.132 |
0.130 |
0.094 |
0.129 |
0.137 |
0.124 |
0.126 |
0.135 |
0.134 |
0.273 |
0.153 |
0.144 |
0.128 |
0.208 |
0.203 |
0.176 |
0.180 |
0.270 |
0.161 |
0.127 |
0.160 |
0.156 |
0.136 |
0.109 |
0.203 |
0.182 |
0.121 |
0.163 |
0.152 |
0.159 |
0.241 |
0.206 |
0.332 |
0.293 |
0.205 |
0.291 |
0.196 |
0.230 |
0.232 |
0.149 |
0.151 |
0.152 |
0.199 |
0.235 |
0.190 |
0.224 |
0.220 |
0.247 |
0.265 |
0.243 |
0.215 |
0.296 |
0.242 |
0.264 |
0.229 |
0.241 |
0.240 |
0.270 |
0.162 |
0.146 |
0.150 |
0.154 |
0.513 |
0.466 |
0.827 |
0.259 |
0.389 |
0.373 |
0.283 |
0.298 |
0.293 |
0.336 |
0.359 |
0.350 |
0.358 |
0.321 |
0.461 |
0.377 |
0.361 |
0.314 |
0.324 |
0.430 |
0.367 |
0.273 |
0.367 |
0.298 |
0.395 |
0.445 |
0.383 |
0.260 |
0.296 |
0.293 |
0.260 |
0.552 |
| F |
— |
0.018 |
0.205 |
0.198 |
— |
0.130 |
0.113 |
— |
— |
0.026 |
0.020 |
— |
— |
— |
0.014 |
0.299 |
0.408 |
0.437 |
0.374 |
— |
— |
0.052 |
— |
— |
— |
— |
— |
0.018 |
— |
0.025 |
— |
— |
0.022 |
— |
— |
0.211 |
0.365 |
— |
0.020 |
0.377 |
0.059 |
0.208 |
— |
— |
0.054 |
0.056 |
0.036 |
— |
0.057 |
0.400 |
0.366 |
— |
0.400 |
0.292 |
— |
— |
— |
0.043 |
0.019 |
0.050 |
— |
0.298 |
— |
0.212 |
0.066 |
0.071 |
0.037 |
— |
0.293 |
— |
— |
0.384 |
0.238 |
— |
0.422 |
0.055 |
0.120 |
0.024 |
— |
0.024 |
0.058 |
0.453 |
0.304 |
— |
0.378 |
0.437 |
— |
— |
0.502 |
0.086 |
0.120 |
0.107 |
— |
0.017 |
| Cl |
0.977 |
0.929 |
1.004 |
1.029 |
0.998 |
0.935 |
0.896 |
0.897 |
0.995 |
1.075 |
1.058 |
1.208 |
1.093 |
1.084 |
1.061 |
1.107 |
1.148 |
1.171 |
1.076 |
1.069 |
1.021 |
1.052 |
0.978 |
1.002 |
1.003 |
0.969 |
0.974 |
1.057 |
1.049 |
0.946 |
0.972 |
0.996 |
1.126 |
1.004 |
1.021 |
1.106 |
1.067 |
1.033 |
1.024 |
1.081 |
1.000 |
1.100 |
1.066 |
1.037 |
1.040 |
1.122 |
1.137 |
1.079 |
1.104 |
1.043 |
1.118 |
0.706 |
1.123 |
1.125 |
1.049 |
1.063 |
1.000 |
0.851 |
1.041 |
1.001 |
1.061 |
1.082 |
0.659 |
0.750 |
0.624 |
0.508 |
0.592 |
0.635 |
0.563 |
0.651 |
0.646 |
0.714 |
0.661 |
0.742 |
0.652 |
0.655 |
0.398 |
0.472 |
0.572 |
0.620 |
0.597 |
0.554 |
0.639 |
0.761 |
0.611 |
0.621 |
0.630 |
0.686 |
0.581 |
0.630 |
0.713 |
0.678 |
0.627 |
0.571 |
|
附表 1 赛马岩体异霞正长岩各阶段异性石电子探针成分(wt%)
Appendix Table 1 Electron-microprobe results for different eudialyte from the lujavrite of the Saima complex (wt%)
|
表 1
Table 1
表 1(Table 1)

表 1
赛马岩体异霞正长岩各阶段异性石代表性电子探针成分(wt%)
Table 1
Representative electron-microprobe results for different eudialyte from the lujavrite of the Saima complex (wt%)
| 产状 |
|
早期异性石核部 |
|
早期异性石边部 |
|
晚期异性石 |
| 序号 | |
1 |
2 |
3 | |
4 |
5 |
6 | |
7 |
8 |
9 |
10 |
| Nb2O5 | |
0.97 |
0.21 |
0.72 | |
2.16 |
2.17 |
3.64 | |
2.82 |
3.61 |
3.01 |
3.36 |
| Ta2O5 | |
0.04 |
0.21 |
— | |
0.16 |
— |
— | |
— |
— |
0.01 |
0.24 |
| SiO2 | |
53.74 |
54.59 |
53.68 | |
48.01 |
48.58 |
45.53 | |
48.27 |
48.53 |
48.10 |
49.40 |
| TiO2 | |
0.43 |
0.81 |
0.38 | |
0.29 |
0.22 |
0.10 | |
0.75 |
0.45 |
0.62 |
0.36 |
| ZrO2 | |
12.06 |
11.86 |
12.64 | |
11.54 |
11.97 |
11.27 | |
12.16 |
12.14 |
12.20 |
12.09 |
| HfO2 | |
0.18 |
0.18 |
0.11 | |
0.31 |
0.18 |
0.23 | |
0.35 |
0.33 |
0.27 |
0.28 |
| Al2O3 | |
0.11 |
0.22 |
0.15 | |
0.12 |
0.12 |
0.05 | |
0.07 |
0.03 |
0.08 |
0.05 |
| La2O3 | |
0.20 |
0.15 |
0.20 | |
0.39 |
0.34 |
0.59 | |
1.47 |
1.79 |
1.58 |
1.73 |
| Ce2O3 | |
0.46 |
0.34 |
0.42 | |
0.90 |
0.87 |
1.24 | |
2.48 |
3.05 |
2.92 |
2.77 |
| Pr2O3 | |
— |
0.03 |
0.03 | |
0.08 |
0.07 |
0.07 | |
0.18 |
0.21 |
0.21 |
0.25 |
| Nd2O3 | |
0.13 |
0.07 |
0.18 | |
0.19 |
0.23 |
0.34 | |
0.59 |
0.60 |
0.54 |
0.51 |
| Sm2O3 | |
0.03 |
— |
0.01 | |
— |
0.03 |
0.03 | |
0.11 |
0.14 |
0.11 |
0.15 |
| Y2O3 | |
— |
0.06 |
0.09 | |
0.20 |
0.19 |
0.32 | |
0.70 |
0.64 |
0.68 |
0.93 |
| CaO | |
10.85 |
11.24 |
10.19 | |
9.89 |
9.91 |
9.14 | |
8.39 |
8.12 |
7.65 |
8.11 |
| MgO | |
0.03 |
0.05 |
0.05 | |
0.06 |
0.08 |
0.08 | |
0.07 |
0.10 |
0.07 |
0.06 |
| FeO | |
6.46 |
6.73 |
6.71 | |
5.48 |
5.37 |
4.82 | |
2.80 |
3.18 |
3.26 |
3.67 |
| MnO | |
1.38 |
1.20 |
0.96 | |
1.69 |
1.82 |
1.97 | |
3.63 |
3.90 |
3.94 |
3.40 |
| SrO | |
2.35 |
1.82 |
2.43 | |
3.22 |
3.03 |
4.00 | |
3.05 |
2.70 |
2.70 |
2.49 |
| Na2O | |
9.27 |
9.03 |
9.38 | |
11.69 |
11.48 |
10.69 | |
7.47 |
5.72 |
8.06 |
4.88 |
| K2O | |
0.22 |
0.21 |
0.25 | |
0.34 |
0.35 |
0.38 | |
0.51 |
0.55 |
0.53 |
0.55 |
| F | |
— |
0.09 |
0.01 | |
— |
0.01 |
0.03 | |
0.24 |
0.15 |
— |
0.26 |
| Cl | |
1.21 |
1.17 |
1.31 | |
1.16 |
1.17 |
1.19 | |
0.82 |
0.76 |
0.85 |
0.76 |
| F=O | |
— |
0.04 |
0.01 | |
— |
0.01 |
0.01 | |
0.10 |
0.06 |
— |
0.11 |
| Cl=O | |
0.28 |
0.27 |
0.30 | |
0.27 |
0.27 |
0.27 | |
0.19 |
0.18 |
0.20 |
0.18 |
| Total | |
99.82 |
99.93 |
99.60 | |
97.62 |
97.93 |
95.41 | |
96.60 |
96.44 |
97.18 |
96.02 |
| 根据∑(Nb+Si+Ti+Zr+Hf+Al) =29 a.p.f.u计算 |
| Nb | |
0.211 |
0.045 |
0.156 | |
0.515 |
0.511 |
0.902 | |
0.657 |
0.838 |
0.704 |
0.769 |
| Ta | |
0.005 |
0.027 |
— | |
0.022 |
— |
— | |
— |
— |
0.002 |
0.032 |
| Si | |
25.734 |
25.789 |
25.660 | |
25.286 |
25.264 |
24.972 | |
24.902 |
24.889 |
24.887 |
25.037 |
| Ti | |
0.155 |
0.288 |
0.136 | |
0.115 |
0.087 |
0.041 | |
0.290 |
0.172 |
0.240 |
0.137 |
| Zr | |
2.815 |
2.732 |
2.947 | |
2.964 |
3.036 |
3.014 | |
3.059 |
3.035 |
3.077 |
2.988 |
| Hf | |
0.025 |
0.024 |
0.016 | |
0.046 |
0.026 |
0.037 | |
0.051 |
0.048 |
0.040 |
0.040 |
| Al | |
0.061 |
0.122 |
0.085 | |
0.074 |
0.075 |
0.035 | |
0.041 |
0.019 |
0.052 |
0.030 |
| La | |
0.035 |
0.025 |
0.036 | |
0.076 |
0.066 |
0.118 | |
0.279 |
0.338 |
0.302 |
0.324 |
| Ce | |
0.080 |
0.059 |
0.074 | |
0.173 |
0.167 |
0.248 | |
0.468 |
0.572 |
0.553 |
0.514 |
| Pr | |
— |
0.005 |
0.005 | |
0.016 |
0.013 |
0.013 | |
0.034 |
0.038 |
0.039 |
0.047 |
| Nd | |
0.022 |
0.012 |
0.031 | |
0.036 |
0.044 |
0.067 | |
0.108 |
0.109 |
0.100 |
0.092 |
| Sm | |
0.005 |
— |
0.002 | |
— |
0.005 |
0.005 | |
0.019 |
0.024 |
0.020 |
0.026 |
| Y | |
— |
0.014 |
0.024 | |
0.056 |
0.054 |
0.093 | |
0.194 |
0.176 |
0.187 |
0.252 |
| Ca | |
5.568 |
5.688 |
5.217 | |
5.584 |
5.522 |
5.373 | |
4.635 |
4.461 |
4.241 |
4.404 |
| Mg | |
0.019 |
0.033 |
0.039 | |
0.047 |
0.065 |
0.065 | |
0.052 |
0.079 |
0.055 |
0.043 |
| Fe | |
2.589 |
2.658 |
2.681 | |
2.414 |
2.337 |
2.210 | |
1.207 |
1.363 |
1.408 |
1.554 |
| Mn | |
0.562 |
0.482 |
0.387 | |
0.752 |
0.799 |
0.913 | |
1.587 |
1.692 |
1.725 |
1.462 |
| Sr | |
0.653 |
0.497 |
0.675 | |
0.984 |
0.912 |
1.271 | |
0.911 |
0.804 |
0.811 |
0.731 |
| Na | |
8.609 |
8.270 |
8.694 | |
11.933 |
11.576 |
11.365 | |
7.472 |
5.689 |
8.085 |
4.796 |
| K | |
0.132 |
0.124 |
0.153 | |
0.230 |
0.232 |
0.265 | |
0.336 |
0.359 |
0.350 |
0.358 |
| F | |
— |
0.130 |
0.020 | |
— |
0.020 |
0.057 | |
0.384 |
0.238 |
— |
0.422 |
| Cl | |
0.977 |
0.935 |
1.058 | |
1.033 |
1.024 |
1.104 | |
0.714 |
0.661 |
0.742 |
0.652 |
|
表 1 赛马岩体异霞正长岩各阶段异性石代表性电子探针成分(wt%)
Table 1 Representative electron-microprobe results for different eudialyte from the lujavrite of the Saima complex (wt%)
|
4.1.2 岩浆晚期至热液期异性石 同一异霞正长岩样品中,常出现不同于早期异性石的细小异性石颗粒(< 20μm),呈他形分布于霓石集合体的晶间或呈细脉状集合体交代早期矿物(图 2e, f),同一背散射电子像照片中此类异性石亮度明显高于周围早期异性石(图 2f),与两者化学成分的差异有关。从异性石形貌及与周围矿物的共生关系判断,此类异性石形成较晚,可能形成于岩浆作用晚期至热液阶段。
晚期异性石总量约为96%~98%,可能含有电子探针无法检测的OH-、CO32-等阴离子(Schilling et al., 2011)。相比于早期异性石,晚期异性石化学成分上具有更低的Fe(2.63%~4.40% FeOT)、Si(45.47%~50.50% SiO2)、Cl(0.55%~0.85% Cl)含量,更高的Nb(2.12%~4.02% Nb2O5)、REE(含Y,4.26%~6.91% REE2O3)含量,此外Sr含量(2.23%~3.50% SrO)介于早期具环带特征异性石核部与边部之间(表 1、附表 1、图 3)。
4.2 异性石蚀变矿物组合 通过场发射扫描电镜背散射电子像对异性石进行精细研究,我们观察到早期异性石发生一系列的热液蚀变,根据蚀变的强弱程度,可以分为以下三种组合。
4.2.1 异性石+钠锆石+霓石±钠沸石 部分被霓石包裹的异性石裂隙或边缘发生微弱蚀变,被次生钠锆石(Na2Si3O9·2H2O)、霓石(NaFeSi2O6)交代,形成异性石+钠锆石+霓石的组合(图 4a)。钠锆石多以他形,细小颗粒集合体的形式出现(< 20μm),与次生霓石紧密共生。值得注意的是,次生霓石产状不同于早期造岩矿物霓石,一般呈细小、他形集合体分布(< 20μm,图 4b)。除此之外,部分异性石的裂隙或边缘也被钠沸石交代。
化学成分上,钠锆石总量介于91.68%~92.37%,其中含有12.65%~13.72% Na2O和1.25%~2.98% CaO(表 2)。霓石总量介于97.42%~99.59%,不同于造岩矿物霓石(Wu et al., 2016, 0.19%~1.74% ZrO2,~0.05% Nb2O5, 表 3、详见电子版附表 2),该蚀变组合中霓石具有较高的Zr(1.43%~6.03% ZrO2,检测限约200×10-6)和Nb(~0.62% Nb2O5,检测限约350×10-6)含量。
表 2
Table 2
表 2(Table 2)

表 2
赛马岩体异霞正长岩各蚀变组合中钠锆石电子探针成分(wt%)
Table 2
Electron-microprobe results for catapleiite in different alteration associations from the lujavrite of the Saima complex (wt%)
| 产状 |
|
异性石+钠锆石组合 |
|
异性石+钠锆石+锆石+钠沸石±霓石组合 |
|
异性石假晶 |
| 序号 | |
1 |
2 |
3 | |
4 |
5 |
6 |
7 | |
8 |
9 |
10 |
| SiO2 | |
45.31 |
44.95 |
44.86 | |
45.62 |
44.87 |
44.23 |
45.86 | |
45.11 |
45.21 |
45.54 |
| TiO2 | |
0.11 |
0.21 |
0.14 | |
— |
0.21 |
— |
— | |
— |
— |
0.23 |
| ZrO2 | |
29.84 |
30.42 |
30.41 | |
29.45 |
29.63 |
30.12 |
30.47 | |
29.34 |
29.33 |
30.02 |
| HfO2 | |
0.45 |
0.64 |
0.81 | |
0.54 |
0.48 |
0.74 |
0.75 | |
0.35 |
0.45 |
0.62 |
| Al2O3 | |
0.02 |
0.01 |
— | |
0.05 |
0.14 |
— |
— | |
— |
0.08 |
— |
| CaO | |
1.25 |
2.62 |
2.98 | |
1.85 |
2.23 |
2.98 |
3.12 | |
4.07 |
3.86 |
3.54 |
| MgO | |
— |
0.01 |
— | |
— |
— |
0.01 |
0.02 | |
— |
0.04 |
— |
| FeO | |
0.68 |
0.27 |
0.21 | |
0.45 |
0.62 |
0.86 |
0.62 | |
0.63 |
0.52 |
0.34 |
| MnO | |
0.18 |
0.21 |
0.17 | |
0.24 |
0.17 |
0.14 |
0.14 | |
0.25 |
0.31 |
0.17 |
| SrO | |
0.04 |
0.02 |
0.04 | |
0.03 |
— |
0.05 |
— | |
— |
0.02 |
0.04 |
| Na2O | |
13.72 |
12.68 |
12.65 | |
13.65 |
13.12 |
12.85 |
11.53 | |
10.14 |
11.45 |
12.03 |
| K2O | |
0.08 |
0.08 |
0.10 | |
0.09 |
0.11 |
— |
0.12 | |
0.09 |
0.07 |
0.10 |
| Total | |
91.68 |
92.12 |
92.37 | |
91.97 |
91.58 |
91.98 |
92.63 | |
89.98 |
91.34 |
92.63 |
| 以9个氧原子计算 |
| Si | |
2.890 |
2.929 |
2.933 | |
2.933 |
2.874 |
2.804 |
2.900 | |
2.915 |
2.910 |
2.930 |
| Ti | |
0.005 |
0.010 |
0.007 | |
— |
0.010 |
— |
— | |
— |
— |
0.011 |
| Zr | |
0.928 |
0.967 |
0.969 | |
0.923 |
0.926 |
0.931 |
0.940 | |
0.924 |
0.921 |
0.942 |
| Hf | |
0.008 |
0.012 |
0.015 | |
0.010 |
0.009 |
0.013 |
0.014 | |
0.006 |
0.008 |
0.011 |
| Al | |
0.002 |
0.001 |
— | |
0.004 |
0.011 |
— |
— | |
— |
0.006 |
— |
| Ca | |
0.085 |
0.183 |
0.209 | |
0.127 |
0.153 |
0.202 |
0.211 | |
0.282 |
0.266 |
0.244 |
| Mg | |
— |
0.001 |
— | |
— |
— |
0.001 |
0.002 | |
— |
0.004 |
— |
| Fe | |
0.036 |
0.015 |
0.011 | |
0.024 |
0.033 |
0.046 |
0.033 | |
0.034 |
0.028 |
0.018 |
| Mn | |
0.010 |
0.012 |
0.009 | |
0.013 |
0.009 |
0.008 |
0.007 | |
0.014 |
0.017 |
0.009 |
| Sr | |
0.001 |
0.001 |
0.002 | |
0.001 |
— |
0.002 |
— | |
— |
0.001 |
0.001 |
| Na | |
1.696 |
1.602 |
1.603 | |
1.701 |
1.629 |
1.579 |
1.413 | |
1.270 |
1.429 |
1.500 |
| K | |
0.007 |
0.007 |
0.008 | |
0.007 |
0.009 |
— |
0.010 | |
0.007 |
0.006 |
0.008 |
| Sum | |
5.669 |
5.738 |
5.767 | |
5.744 |
5.663 |
5.585 |
5.530 | |
5.453 |
5.595 |
5.677 |
|
表 2 赛马岩体异霞正长岩各蚀变组合中钠锆石电子探针成分(wt%)
Table 2 Electron-microprobe results for catapleiite in different alteration associations from the lujavrite of the Saima complex (wt%)
|
附表 2
Appendix Table 2
附表 2(Appendix Table 2)

附表 2
赛马岩体异霞正长岩各阶段霓石代表性电子探针成分(wt%)
Appendix Table 2
Electron-microprobe results for different aegirine from the lujavrite of the Saima complex (wt%)
| 产状 |
|
造岩矿物霓石 |
|
异性石+钠锆石+霓石±钠沸石蚀变组合中霓石 |
|
异性石假晶中霓石 |
| 序号 |
|
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
|
11 |
12 |
13 |
14 |
15 |
16 |
17 |
18 |
19 |
20 |
21 |
23 |
24 |
25 |
|
26 |
27 |
28 |
29 |
30 |
31 |
32 |
33 |
34 |
35 |
36 |
37 |
38 |
39 |
| Nb2O5 |
|
0.06 |
— |
0.07 |
— |
0.05 |
— |
0.03 |
— |
0.02 |
0.03 |
|
0.43 |
0.03 |
0.01 |
0.46 |
0.62 |
0.62 |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
|
0.76 |
0.59 |
0.75 |
0.99 |
0.75 |
0.88 |
1.06 |
0.86 |
0.16 |
0.78 |
n.d. |
n.d. |
n.d. |
n.d. |
| SiO2 |
52.01 |
51.74 |
51.81 |
52.79 |
52.31 |
52.21 |
51.70 |
51.62 |
52.08 |
52.44 |
50.93 |
52.06 |
51.65 |
50.53 |
51.02 |
51.00 |
52.09 |
52.99 |
51.33 |
51.51 |
51.12 |
52.16 |
51.05 |
51.41 |
51.30 |
51.23 |
51.76 |
51.13 |
50.65 |
50.79 |
50.83 |
51.09 |
50.87 |
50.98 |
51.26 |
51.91 |
51.55 |
51.47 |
| TiO2 |
1.20 |
1.43 |
1.13 |
1.45 |
1.38 |
1.09 |
1.58 |
1.16 |
1.76 |
1.06 |
2.23 |
1.85 |
1.52 |
2.38 |
2.31 |
2.12 |
1.23 |
2.48 |
1.83 |
2.10 |
3.60 |
2.29 |
1.91 |
2.82 |
1.74 |
2.20 |
1.84 |
2.13 |
2.02 |
2.14 |
1.73 |
2.11 |
2.48 |
2.65 |
1.94 |
1.86 |
1.86 |
1.98 |
| ZrO2 |
0.34 |
0.48 |
0.28 |
0.33 |
0.35 |
0.35 |
0.54 |
0.27 |
0.48 |
0.43 |
4.63 |
4.18 |
4.63 |
6.03 |
3.67 |
5.51 |
3.20 |
1.43 |
3.87 |
5.04 |
4.03 |
2.60 |
5.73 |
4.54 |
2.78 |
2.83 |
5.47 |
5.02 |
5.96 |
2.14 |
2.56 |
4.29 |
4.66 |
4.19 |
4.30 |
4.83 |
3.03 |
5.37 |
| Al2O3 |
0.78 |
0.66 |
0.78 |
0.95 |
0.86 |
0.76 |
0.66 |
0.68 |
0.85 |
0.79 |
0.33 |
0.10 |
0.11 |
0.21 |
0.34 |
0.21 |
0.85 |
0.42 |
0.29 |
0.35 |
0.15 |
0.47 |
0.17 |
0.23 |
0.16 |
0.64 |
0.11 |
0.51 |
0.11 |
0.39 |
0.22 |
0.13 |
0.18 |
0.19 |
0.28 |
0.08 |
0.15 |
0.08 |
| Cr2O3 |
— |
0.01 |
0.01 |
— |
0.03 |
— |
0.02 |
0.05 |
0.02 |
— |
— |
— |
0.01 |
0.02 |
— |
0.01 |
— |
— |
0.04 |
0.04 |
0.05 |
0.01 |
— |
0.01 |
— |
— |
— |
0.02 |
0.04 |
0.01 |
— |
— |
— |
— |
— |
— |
— |
0.00 |
| MgO |
1.07 |
0.91 |
0.89 |
1.15 |
1.49 |
2.06 |
1.11 |
2.10 |
2.19 |
2.37 |
0.21 |
0.14 |
0.16 |
0.28 |
0.21 |
0.17 |
1.71 |
0.18 |
0.13 |
0.21 |
0.18 |
0.55 |
0.23 |
0.27 |
0.26 |
0.85 |
0.28 |
0.62 |
0.23 |
0.28 |
0.16 |
0.18 |
0.17 |
0.18 |
0.34 |
0.17 |
0.24 |
0.19 |
| CaO |
3.26 |
3.42 |
3.01 |
2.99 |
3.48 |
5.45 |
4.17 |
5.24 |
5.70 |
5.75 |
0.98 |
0.07 |
0.10 |
1.19 |
0.74 |
0.43 |
3.74 |
0.18 |
1.69 |
0.99 |
1.19 |
1.77 |
1.36 |
0.66 |
0.54 |
0.52 |
0.51 |
0.30 |
0.38 |
0.84 |
0.68 |
0.41 |
1.12 |
0.82 |
0.41 |
0.71 |
0.43 |
0.32 |
| FeO |
27.53 |
27.31 |
27.56 |
26.39 |
26.63 |
25.65 |
26.46 |
25.41 |
24.78 |
24.96 |
22.79 |
25.45 |
25.81 |
21.61 |
23.74 |
22.84 |
24.40 |
27.08 |
22.70 |
22.86 |
22.06 |
23.38 |
21.30 |
22.60 |
24.47 |
22.67 |
22.07 |
21.44 |
21.47 |
23.79 |
24.77 |
23.15 |
21.03 |
21.95 |
22.01 |
21.71 |
24.18 |
21.72 |
| MnO |
0.21 |
0.21 |
0.16 |
0.22 |
0.30 |
0.31 |
0.25 |
0.27 |
0.38 |
0.34 |
1.87 |
0.92 |
0.90 |
2.48 |
2.01 |
1.86 |
0.37 |
0.59 |
3.51 |
1.99 |
2.05 |
2.57 |
2.62 |
2.24 |
1.96 |
2.37 |
2.48 |
2.30 |
2.67 |
2.13 |
1.96 |
1.61 |
2.42 |
2.54 |
2.61 |
2.63 |
1.88 |
2.71 |
| Na2O |
11.84 |
11.83 |
12.10 |
12.44 |
11.82 |
10.74 |
11.23 |
10.06 |
10.60 |
10.43 |
13.40 |
13.98 |
13.89 |
13.07 |
13.36 |
13.59 |
11.77 |
14.26 |
12.93 |
13.58 |
13.18 |
12.97 |
13.07 |
13.84 |
13.46 |
13.41 |
12.67 |
13.76 |
13.55 |
13.16 |
13.25 |
13.47 |
13.08 |
13.58 |
13.20 |
13.17 |
13.60 |
13.92 |
| Total |
98.30 |
98.00 |
97.81 |
98.71 |
98.72 |
98.62 |
97.75 |
96.86 |
98.86 |
98.60 |
97.82 |
98.77 |
98.78 |
98.26 |
98.00 |
98.34 |
99.37 |
99.59 |
98.31 |
98.66 |
97.61 |
98.77 |
97.42 |
98.63 |
97.44 |
97.31 |
97.94 |
98.21 |
97.82 |
96.53 |
97.22 |
97.29 |
96.18 |
97.85 |
96.36 |
97.09 |
96.91 |
97.75 |
| 以4个阳离子计算 |
| Nb |
0.001 |
— |
0.001 |
— |
0.001 |
— |
— |
— |
— |
— |
0.008 |
0.001 |
— |
0.008 |
0.011 |
0.011 |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
n.d. |
0.013 |
0.010 |
0.013 |
0.017 |
0.013 |
0.015 |
0.018 |
0.015 |
0.003 |
0.014 |
n.d. |
n.d. |
n.d. |
n.d. |
| Si |
1.976 |
1.974 |
1.974 |
1.983 |
1.974 |
1.979 |
1.985 |
2.001 |
1.972 |
1.989 |
1.969 |
1.982 |
1.971 |
1.963 |
1.966 |
1.970 |
1.972 |
1.975 |
1.974 |
1.972 |
1.980 |
1.981 |
1.989 |
1.963 |
1.979 |
1.969 |
2.022 |
1.966 |
1.971 |
1.976 |
1.971 |
1.987 |
2.000 |
1.969 |
2.004 |
2.021 |
1.991 |
1.984 |
| Ti |
0.034 |
0.041 |
0.033 |
0.041 |
0.039 |
0.031 |
0.046 |
0.034 |
0.050 |
0.030 |
0.065 |
0.053 |
0.044 |
0.070 |
0.067 |
0.061 |
0.035 |
0.070 |
0.053 |
0.060 |
0.105 |
0.065 |
0.056 |
0.081 |
0.050 |
0.064 |
0.054 |
0.062 |
0.059 |
0.063 |
0.050 |
0.062 |
0.073 |
0.077 |
0.057 |
0.055 |
0.054 |
0.057 |
| Zr |
0.006 |
0.009 |
0.005 |
0.006 |
0.007 |
0.007 |
0.010 |
0.005 |
0.009 |
0.008 |
0.087 |
0.078 |
0.086 |
0.114 |
0.069 |
0.104 |
0.059 |
0.026 |
0.072 |
0.094 |
0.076 |
0.048 |
0.109 |
0.084 |
0.052 |
0.053 |
0.104 |
0.094 |
0.113 |
0.041 |
0.048 |
0.081 |
0.089 |
0.079 |
0.082 |
0.092 |
0.057 |
0.101 |
| Al |
0.035 |
0.030 |
0.035 |
0.042 |
0.038 |
0.034 |
0.030 |
0.031 |
0.038 |
0.035 |
0.015 |
0.004 |
0.005 |
0.010 |
0.015 |
0.010 |
0.038 |
0.018 |
0.013 |
0.016 |
0.007 |
0.021 |
0.008 |
0.010 |
0.007 |
0.029 |
0.005 |
0.023 |
0.005 |
0.018 |
0.010 |
0.006 |
0.008 |
0.009 |
0.013 |
0.004 |
0.007 |
0.004 |
| Cr |
— |
— |
— |
— |
0.001 |
— |
0.001 |
0.001 |
0.001 |
— |
— |
— |
— |
0.001 |
— |
— |
— |
— |
0.001 |
0.001 |
0.002 |
— |
— |
0.000 |
— |
— |
— |
— |
0.001 |
— |
— |
— |
— |
— |
— |
— |
— |
— |
| Mg |
0.061 |
0.052 |
0.050 |
0.064 |
0.084 |
0.116 |
0.064 |
0.122 |
0.123 |
0.134 |
0.012 |
0.008 |
0.009 |
0.016 |
0.012 |
0.010 |
0.097 |
0.010 |
0.007 |
0.012 |
0.010 |
0.031 |
0.013 |
0.015 |
0.015 |
0.049 |
0.016 |
0.035 |
0.013 |
0.016 |
0.009 |
0.010 |
0.010 |
0.010 |
0.020 |
0.010 |
0.014 |
0.011 |
| Ca |
0.133 |
0.140 |
0.123 |
0.120 |
0.141 |
0.221 |
0.171 |
0.218 |
0.231 |
0.234 |
0.041 |
0.003 |
0.004 |
0.050 |
0.030 |
0.018 |
0.152 |
0.007 |
0.070 |
0.041 |
0.049 |
0.072 |
0.057 |
0.027 |
0.022 |
0.021 |
0.021 |
0.012 |
0.016 |
0.035 |
0.028 |
0.017 |
0.047 |
0.034 |
0.017 |
0.030 |
0.018 |
0.013 |
| Fe |
0.875 |
0.872 |
0.879 |
0.829 |
0.841 |
0.813 |
0.850 |
0.824 |
0.785 |
0.792 |
0.737 |
0.810 |
0.824 |
0.702 |
0.765 |
0.738 |
0.772 |
0.844 |
0.730 |
0.732 |
0.714 |
0.743 |
0.694 |
0.722 |
0.789 |
0.729 |
0.721 |
0.689 |
0.699 |
0.774 |
0.803 |
0.753 |
0.691 |
0.709 |
0.720 |
0.707 |
0.781 |
0.700 |
| Mn |
0.007 |
0.007 |
0.005 |
0.007 |
0.010 |
0.010 |
0.008 |
0.009 |
0.012 |
0.011 |
0.061 |
0.030 |
0.029 |
0.081 |
0.066 |
0.061 |
0.012 |
0.019 |
0.114 |
0.065 |
0.067 |
0.083 |
0.086 |
0.073 |
0.064 |
0.077 |
0.082 |
0.075 |
0.088 |
0.070 |
0.064 |
0.053 |
0.081 |
0.083 |
0.087 |
0.087 |
0.062 |
0.088 |
| Na |
0.872 |
0.876 |
0.894 |
0.906 |
0.865 |
0.789 |
0.836 |
0.756 |
0.778 |
0.767 |
1.005 |
1.032 |
1.028 |
0.985 |
0.998 |
1.018 |
0.864 |
1.031 |
0.964 |
1.008 |
0.989 |
0.955 |
0.987 |
1.024 |
1.007 |
1.000 |
0.960 |
1.026 |
1.022 |
0.993 |
0.996 |
1.016 |
0.997 |
1.017 |
1.001 |
0.995 |
1.018 |
1.041 |
| Sum |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
4.000 |
| 注:n.d.=not detected |
|
附表 2 赛马岩体异霞正长岩各阶段霓石代表性电子探针成分(wt%)
Appendix Table 2 Electron-microprobe results for different aegirine from the lujavrite of the Saima complex (wt%)
|
表 3
Table 3
表 3(Table 3)

表 3
赛马岩体异霞正长岩各阶段霓石代表性电子探针成分(wt%)
Table 3
Representative electron-microprobe results for different aegirine from the lujavrite of the Saima complex (wt%)
| 产状 |
|
造岩矿物 |
|
异性石+钠锆石+霓石±钠沸石蚀变组合 |
|
异性石假晶 |
| 序号 | |
1 |
2 |
3 | |
4 |
5 |
6 |
7 | |
8 |
9 |
10 |
11 |
| Nb2O5 | |
0.06 |
— |
0.07 | |
0.43 |
0.03 |
0.01 |
0.46 | |
0.76 |
0.59 |
0.75 |
0.99 |
| SiO2 | |
52.01 |
51.74 |
51.81 | |
50.93 |
52.06 |
51.65 |
50.53 | |
51.30 |
51.23 |
51.76 |
51.13 |
| TiO2 | |
1.20 |
1.43 |
1.13 | |
2.23 |
1.85 |
1.52 |
2.38 | |
1.74 |
2.20 |
1.84 |
2.13 |
| ZrO2 | |
0.34 |
0.48 |
0.28 | |
4.63 |
4.18 |
4.63 |
6.03 | |
2.78 |
2.83 |
5.47 |
5.02 |
| Al2O3 | |
0.78 |
0.66 |
0.78 | |
0.33 |
0.10 |
0.11 |
0.21 | |
0.16 |
0.64 |
0.11 |
0.51 |
| Cr2O3 | |
— |
0.01 |
0.01 | |
— |
— |
0.01 |
0.02 | |
— |
— |
— |
0.02 |
| MgO | |
1.07 |
0.91 |
0.89 | |
0.21 |
0.14 |
0.16 |
0.28 | |
0.26 |
0.85 |
0.28 |
0.62 |
| CaO | |
3.26 |
3.42 |
3.01 | |
0.98 |
0.07 |
0.10 |
1.19 | |
0.54 |
0.52 |
0.51 |
0.30 |
| FeO | |
27.53 |
27.31 |
27.56 | |
22.79 |
25.45 |
25.81 |
21.61 | |
24.47 |
22.67 |
22.07 |
21.44 |
| MnO | |
0.21 |
0.21 |
0.16 | |
1.87 |
0.92 |
0.90 |
2.48 | |
1.96 |
2.37 |
2.48 |
2.30 |
| Na2O | |
11.84 |
11.83 |
12.10 | |
13.40 |
13.98 |
13.89 |
13.07 | |
13.46 |
13.41 |
12.67 |
13.76 |
| Total | |
98.30 |
98.00 |
97.81 | |
97.82 |
98.77 |
98.78 |
98.26 | |
97.44 |
97.31 |
97.94 |
98.21 |
| 以4个阳离子计算 |
| Nb | |
0.001 |
— |
0.001 | |
0.008 |
0.001 |
— |
0.008 | |
0.013 |
0.010 |
0.013 |
0.017 |
| Si | |
1.976 |
1.974 |
1.974 | |
1.969 |
1.982 |
1.971 |
1.963 | |
1.979 |
1.969 |
2.022 |
1.966 |
| Ti | |
0.034 |
0.041 |
0.033 | |
0.065 |
0.053 |
0.044 |
0.070 | |
0.050 |
0.064 |
0.054 |
0.062 |
| Zr | |
0.006 |
0.009 |
0.005 | |
0.087 |
0.078 |
0.086 |
0.114 | |
0.052 |
0.053 |
0.104 |
0.094 |
| Al | |
0.035 |
0.030 |
0.035 | |
0.015 |
0.004 |
0.005 |
0.010 | |
0.007 |
0.029 |
0.005 |
0.023 |
| Cr | |
— |
— |
— | |
— |
— |
— |
0.001 | |
— |
— |
— |
— |
| Mg | |
0.061 |
0.052 |
0.050 | |
0.012 |
0.008 |
0.009 |
0.016 | |
0.015 |
0.049 |
0.016 |
0.035 |
| Ca | |
0.133 |
0.140 |
0.123 | |
0.041 |
0.003 |
0.004 |
0.050 | |
0.022 |
0.021 |
0.021 |
0.012 |
| Fe | |
0.875 |
0.872 |
0.879 | |
0.737 |
0.810 |
0.824 |
0.702 | |
0.789 |
0.729 |
0.721 |
0.689 |
| Mn | |
0.007 |
0.007 |
0.005 | |
0.061 |
0.030 |
0.029 |
0.081 | |
0.064 |
0.077 |
0.082 |
0.075 |
| Na | |
0.872 |
0.876 |
0.894 | |
1.005 |
1.032 |
1.028 |
0.985 | |
1.007 |
1.000 |
0.960 |
1.026 |
| Sum | |
4.000 |
4.000 |
4.000 | |
4.000 |
4.000 |
4.000 |
4.000 | |
4.000 |
4.000 |
4.000 |
4.000 |
|
表 3 赛马岩体异霞正长岩各阶段霓石代表性电子探针成分(wt%)
Table 3 Representative electron-microprobe results for different aegirine from the lujavrite of the Saima complex (wt%)
|
4.2.2 异性石+钠锆石+锆石+钠沸石±霓石 我们进一步观察到钾长石、霞石发生钠沸石化,而与之共生的异性石发生更为强烈的蚀变(图 4c),尽管大部分异性石核部保持新鲜,但边部形成异性石+钠锆石+锆石+钠沸石的组合(图 4d)。其中钠锆石和锆石粒径 < 5μm,多呈细脉状或他形粒状分布于异性石边缘或被钠沸石包裹,部分细小锆石颗粒分布于钠锆石的周围。图 4d中异性石边缘具溶蚀明显的港湾状特征,且被包裹的霓石呈半自形粒状,粒径>20μm,为早期造岩矿物。
钠锆石化学成分与组合(1)中钠锆石相似,总量约为92%,其中含有12.85%~13.65% Na2O和1.85%~2.98% CaO(表 2);锆石化学成分与其他碱性岩体中异性石蚀变组合次生锆石相似(Coulson, 1997; Mitchell and Liferovich, 2006; Karup-Møller et al., 2010),总量94.77%~98.00%,可能含有大量的OH-等电子探针无法测量的成分,且含有较高的Al(0.79%~3.30% Al2O3,检测限约160×10-6)、Fe(0.50%~0.88% FeOT,检测限约300×10-6)和Ca(0.61%~1.11% CaO,检测限约150×10-6,表 4、详见电子版附表 3、图 5)含量,其中Ca含量整体低于该岩体钾锆石蚀变组合中次生锆石Ca含量(0.92%~1.45% CaO, Wu et al., 2015)。钠沸石是碱性岩中典型的次生含水矿物,该组合中钠沸石总量约为90%,主要含有14.68%~16.66% Na2O,25.86%~27.98% Al2O3,46.20%~47.28% SiO2及少量CaO(表 5)。
表 4
Table 4
表 4(Table 4)

表 4
赛马岩体异霞正长岩异性石蚀变组合中锆石代表性电子探针成分(wt%)
Table 4
Representative electron-microprobe results for zircon in different alteration associations from the lujavrite of the Saima complex (wt%)
| 产状 |
|
异性石+钠锆石+锆石+钠沸石±霓石组合 |
|
异性石假晶 |
| 序号 | |
1 |
2 |
3 |
4 |
5 | |
6 |
7 |
8 |
9 |
10 |
| SiO2 | |
31.19 |
31.46 |
31.36 |
30.53 |
29.84 | |
30.62 |
30.27 |
30.89 |
30.80 |
30.91 |
| TiO2 | |
0.12 |
0.26 |
0.26 |
0.21 |
0.36 | |
0.29 |
0.15 |
0.14 |
0.16 |
0.19 |
| ZrO2 | |
61.58 |
61.88 |
62.85 |
64.27 |
61.38 | |
62.26 |
61.98 |
63.37 |
60.94 |
62.86 |
| HfO2 | |
0.98 |
0.62 |
0.99 |
1.20 |
0.70 | |
1.29 |
1.00 |
1.31 |
0.96 |
1.23 |
| UO2 | |
0.04 |
— |
— |
— |
0.28 | |
— |
— |
— |
0.02 |
— |
| ThO2 | |
0.14 |
0.35 |
0.30 |
— |
0.97 | |
0.03 |
0.17 |
0.03 |
— |
0.12 |
| Al2O3 | |
1.18 |
0.99 |
0.79 |
0.89 |
1.42 | |
0.89 |
1.10 |
0.67 |
1.35 |
0.74 |
| Y2O3 | |
0.78 |
0.59 |
0.50 |
0.65 |
1.28 | |
0.55 |
1.02 |
0.70 |
1.14 |
0.67 |
| FeO | |
0.86 |
0.75 |
0.62 |
0.44 |
0.82 | |
0.69 |
1.16 |
0.59 |
0.47 |
0.58 |
| CaO | |
0.91 |
1.07 |
0.85 |
0.44 |
1.11 | |
0.59 |
0.42 |
0.56 |
0.71 |
0.68 |
| PbO | |
0.08 |
0.33 |
0.24 |
— |
0.50 | |
0.04 |
0.03 |
— |
— |
— |
| K2O | |
0.04 |
— |
0.02 |
0.04 |
0.03 | |
0.04 |
0.05 |
0.03 |
0.03 |
0.00 |
| Na2O | |
0.10 |
0.09 |
0.18 |
0.19 |
0.08 | |
0.13 |
0.16 |
0.14 |
0.15 |
0.08 |
| Total | |
98.00 |
98.38 |
98.95 |
98.84 |
98.78 | |
97.41 |
97.52 |
98.41 |
96.71 |
98.06 |
| 以4个氧原子计算 |
| Si | |
0.976 |
0.980 |
0.975 |
0.955 |
0.942 | |
0.968 |
0.959 |
0.969 |
0.974 |
0.971 |
| Ti | |
0.003 |
0.006 |
0.006 |
0.005 |
0.009 | |
0.007 |
0.004 |
0.003 |
0.004 |
0.004 |
| Zr | |
0.940 |
0.940 |
0.953 |
0.980 |
0.945 | |
0.960 |
0.958 |
0.969 |
0.940 |
0.963 |
| Hf | |
0.009 |
0.006 |
0.009 |
0.011 |
0.006 | |
0.012 |
0.009 |
0.012 |
0.009 |
0.011 |
| U | |
— |
— |
— |
— |
0.002 | |
— |
— |
— |
— |
— |
| Th | |
0.001 |
0.002 |
0.002 |
— |
0.007 | |
— |
0.001 |
— |
— |
0.001 |
| Al | |
0.044 |
0.036 |
0.029 |
0.033 |
0.053 | |
0.033 |
0.041 |
0.025 |
0.050 |
0.027 |
| Y | |
0.013 |
0.010 |
0.008 |
0.011 |
0.021 | |
0.009 |
0.017 |
0.012 |
0.019 |
0.011 |
| Fe | |
0.023 |
0.019 |
0.016 |
0.011 |
0.022 | |
0.018 |
0.031 |
0.015 |
0.012 |
0.015 |
| Ca | |
0.031 |
0.036 |
0.028 |
0.015 |
0.038 | |
0.020 |
0.014 |
0.019 |
0.024 |
0.023 |
| Pb | |
0.001 |
0.003 |
0.002 |
— |
0.004 | |
— |
— |
— |
— |
— |
| K | |
0.002 |
— |
0.001 |
0.001 |
0.001 | |
0.002 |
0.002 |
0.001 |
0.001 |
— |
| Na | |
0.006 |
0.005 |
0.011 |
0.011 |
0.005 | |
0.008 |
0.010 |
0.009 |
0.009 |
0.005 |
| Sum | |
2.047 |
2.043 |
2.040 |
2.033 |
2.055 | |
2.037 |
2.046 |
2.034 |
2.042 |
2.031 |
|
表 4 赛马岩体异霞正长岩异性石蚀变组合中锆石代表性电子探针成分(wt%)
Table 4 Representative electron-microprobe results for zircon in different alteration associations from the lujavrite of the Saima complex (wt%)
|
附表 3
Appendix Table 3
附表 3(Appendix Table 3)

附表 3
赛马岩体异霞正长岩异性石蚀变组合中锆石电子探针成分(wt%)
Appendix Table 3
Electron-microprobe results for zircon in different alteration associations from the lujavrite of the Saima complex (wt%)
| 产状 |
|
异性石+钠锆石+锆石+钠沸石±霓石组合中锆石 |
|
异性石假晶中锆石 |
| 序号 |
|
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
12 |
13 |
14 |
15 |
16 |
17 |
18 |
19 |
|
20 |
21 |
22 |
23 |
24 |
25 |
26 |
27 |
28 |
29 |
30 |
31 |
32 |
33 |
34 |
35 |
36 |
37 |
38 |
39 |
40 |
| SiO2 |
|
30.54 |
30.82 |
31.19 |
29.76 |
30.89 |
31.56 |
33.90 |
32.49 |
31.46 |
31.36 |
30.53 |
30.38 |
25.55 |
31.34 |
31.29 |
30.06 |
28.82 |
30.20 |
29.84 |
|
30.33 |
30.11 |
29.04 |
30.62 |
30.27 |
30.89 |
30.80 |
30.91 |
30.87 |
32.26 |
30.41 |
28.18 |
29.58 |
30.64 |
30.92 |
31.00 |
30.87 |
31.04 |
30.09 |
30.82 |
31.16 |
| TiO2 |
0.25 |
0.15 |
0.12 |
0.24 |
0.23 |
0.20 |
0.30 |
0.45 |
0.26 |
0.26 |
0.21 |
0.22 |
0.48 |
0.34 |
0.09 |
0.15 |
0.48 |
0.32 |
0.36 |
0.22 |
0.14 |
0.09 |
0.29 |
0.15 |
0.14 |
0.16 |
0.19 |
0.16 |
0.18 |
0.09 |
0.28 |
0.16 |
0.23 |
0.19 |
0.19 |
0.18 |
0.20 |
0.17 |
0.17 |
0.32 |
| ZrO2 |
61.24 |
59.85 |
61.58 |
58.81 |
61.99 |
61.40 |
57.92 |
56.19 |
61.88 |
62.85 |
64.27 |
62.60 |
60.89 |
61.12 |
60.90 |
61.97 |
57.62 |
59.50 |
61.38 |
60.02 |
62.76 |
63.26 |
62.26 |
61.98 |
63.37 |
60.94 |
62.86 |
62.67 |
59.09 |
59.70 |
61.95 |
64.91 |
63.52 |
60.92 |
63.04 |
64.10 |
63.47 |
62.35 |
61.60 |
65.53 |
| HfO2 |
1.04 |
1.12 |
0.98 |
1.34 |
1.14 |
1.06 |
0.87 |
0.78 |
0.62 |
0.99 |
1.20 |
1.09 |
1.04 |
1.01 |
1.27 |
1.35 |
0.66 |
0.90 |
0.70 |
0.81 |
1.00 |
1.10 |
1.29 |
1.00 |
1.31 |
0.96 |
1.23 |
1.10 |
0.82 |
1.79 |
1.11 |
0.62 |
1.34 |
1.16 |
1.01 |
1.36 |
0.86 |
0.91 |
1.00 |
1.08 |
| UO2 |
— |
0.06 |
0.04 |
0.01 |
0.10 |
0.01 |
0.03 |
0.05 |
— |
— |
— |
— |
0.03 |
— |
— |
— |
0.17 |
0.19 |
0.28 |
— |
0.01 |
— |
— |
— |
— |
0.02 |
— |
— |
0.01 |
— |
— |
— |
— |
— |
— |
0.07 |
0.02 |
0.09 |
0.02 |
0.01 |
| ThO2 |
0.06 |
0.16 |
0.14 |
— |
0.10 |
0.12 |
0.05 |
0.20 |
0.35 |
0.30 |
— |
0.06 |
0.07 |
— |
— |
— |
1.63 |
1.30 |
0.97 |
— |
0.18 |
0.03 |
0.03 |
0.17 |
0.03 |
— |
0.12 |
0.01 |
0.03 |
0.20 |
0.05 |
0.16 |
— |
— |
0.06 |
— |
0.09 |
0.30 |
— |
— |
| Al2O3 |
1.00 |
1.02 |
1.18 |
1.99 |
1.13 |
0.94 |
1.66 |
3.30 |
0.99 |
0.79 |
0.89 |
1.44 |
1.74 |
1.15 |
2.93 |
0.87 |
1.26 |
0.86 |
1.42 |
1.49 |
1.13 |
1.02 |
0.89 |
1.10 |
0.67 |
1.35 |
0.74 |
0.75 |
1.11 |
1.64 |
1.75 |
1.81 |
0.67 |
1.02 |
0.81 |
0.97 |
0.93 |
0.77 |
1.24 |
1.12 |
| Y2O3 |
0.59 |
0.79 |
0.78 |
0.81 |
0.80 |
1.28 |
0.63 |
1.06 |
0.59 |
0.50 |
0.65 |
0.55 |
0.32 |
0.59 |
0.32 |
0.55 |
2.09 |
1.46 |
1.28 |
1.03 |
1.17 |
0.44 |
0.55 |
1.02 |
0.70 |
1.14 |
0.67 |
0.78 |
0.60 |
0.14 |
0.59 |
0.25 |
0.65 |
0.84 |
0.83 |
0.56 |
0.83 |
0.71 |
0.61 |
0.30 |
| FeO |
0.50 |
0.56 |
0.86 |
0.88 |
0.57 |
0.56 |
1.26 |
0.99 |
0.75 |
0.62 |
0.44 |
0.39 |
3.70 |
0.58 |
0.42 |
0.57 |
0.81 |
0.97 |
0.82 |
0.48 |
1.10 |
0.97 |
0.69 |
1.16 |
0.59 |
0.47 |
0.58 |
0.65 |
0.47 |
1.18 |
0.50 |
0.41 |
0.55 |
0.59 |
0.75 |
0.66 |
0.63 |
0.39 |
0.43 |
0.68 |
| CaO |
0.88 |
0.85 |
0.91 |
0.61 |
0.68 |
0.56 |
0.77 |
1.00 |
1.07 |
0.85 |
0.44 |
0.62 |
0.65 |
0.77 |
0.64 |
0.62 |
1.10 |
0.91 |
1.11 |
0.60 |
0.49 |
0.48 |
0.59 |
0.42 |
0.56 |
0.71 |
0.68 |
0.78 |
0.95 |
0.79 |
0.58 |
0.67 |
0.69 |
0.87 |
0.81 |
0.50 |
0.90 |
0.73 |
0.88 |
0.95 |
| PbO |
0.03 |
0.04 |
0.08 |
0.01 |
0.08 |
— |
— |
0.46 |
0.33 |
0.24 |
— |
— |
0.00 |
— |
— |
0.43 |
0.81 |
0.57 |
0.50 |
— |
0.07 |
— |
0.04 |
0.03 |
— |
— |
— |
0.03 |
— |
— |
0.09 |
— |
0.03 |
— |
0.03 |
0.08 |
0.29 |
0.19 |
0.41 |
— |
| K2O |
0.05 |
0.03 |
0.04 |
0.07 |
0.03 |
— |
0.06 |
— |
— |
0.02 |
0.04 |
0.07 |
0.03 |
0.06 |
0.03 |
0.02 |
0.01 |
0.03 |
0.03 |
0.07 |
0.07 |
0.02 |
0.04 |
0.05 |
0.03 |
0.03 |
0.00 |
0.04 |
0.03 |
— |
0.03 |
0.03 |
0.04 |
0.06 |
0.03 |
0.06 |
0.02 |
0.04 |
0.04 |
0.03 |
| Na2O |
0.07 |
0.14 |
0.10 |
0.26 |
0.14 |
0.11 |
0.29 |
0.60 |
0.09 |
0.18 |
0.19 |
0.27 |
0.79 |
0.44 |
0.82 |
0.15 |
0.05 |
0.17 |
0.08 |
0.20 |
0.24 |
0.51 |
0.13 |
0.16 |
0.14 |
0.15 |
0.08 |
0.17 |
0.06 |
0.33 |
0.62 |
0.62 |
0.06 |
0.22 |
0.90 |
0.21 |
0.08 |
0.13 |
0.10 |
0.11 |
| Total |
96.24 |
95.59 |
98.00 |
94.77 |
97.87 |
97.79 |
97.75 |
97.58 |
98.38 |
98.95 |
98.84 |
97.69 |
95.30 |
97.40 |
98.70 |
96.74 |
95.50 |
97.34 |
98.78 |
95.24 |
98.47 |
96.97 |
97.41 |
97.52 |
98.41 |
96.71 |
98.06 |
98.01 |
95.61 |
96.24 |
95.73 |
99.20 |
98.43 |
96.78 |
99.46 |
99.62 |
99.33 |
96.87 |
97.32 |
101.29 |
| 以4个氧原子计算 |
| Si |
0.973 |
0.987 |
0.976 |
0.961 |
0.970 |
0.987 |
1.036 |
0.997 |
0.980 |
0.975 |
0.955 |
0.956 |
0.853 |
0.982 |
0.961 |
0.962 |
0.947 |
0.967 |
0.942 |
0.973 |
0.949 |
0.933 |
0.968 |
0.959 |
0.969 |
0.974 |
0.971 |
0.970 |
1.017 |
0.970 |
0.916 |
0.923 |
0.962 |
0.979 |
0.962 |
0.958 |
0.964 |
0.961 |
0.972 |
0.950 |
| Ti |
0.006 |
0.004 |
0.003 |
0.006 |
0.005 |
0.005 |
0.007 |
0.010 |
0.006 |
0.006 |
0.005 |
0.005 |
0.012 |
0.008 |
0.002 |
0.004 |
0.012 |
0.008 |
0.009 |
0.005 |
0.003 |
0.002 |
0.007 |
0.004 |
0.003 |
0.004 |
0.004 |
0.004 |
0.004 |
0.002 |
0.007 |
0.004 |
0.005 |
0.005 |
0.004 |
0.004 |
0.005 |
0.004 |
0.004 |
0.007 |
| Zr |
0.952 |
0.934 |
0.940 |
0.926 |
0.950 |
0.937 |
0.863 |
0.841 |
0.940 |
0.953 |
0.980 |
0.961 |
0.992 |
0.934 |
0.912 |
0.967 |
0.923 |
0.929 |
0.945 |
0.939 |
0.964 |
0.991 |
0.960 |
0.958 |
0.969 |
0.940 |
0.963 |
0.961 |
0.909 |
0.928 |
0.982 |
0.987 |
0.973 |
0.941 |
0.954 |
0.970 |
0.961 |
0.972 |
0.948 |
0.974 |
| Hf |
0.009 |
0.010 |
0.009 |
0.012 |
0.010 |
0.009 |
0.008 |
0.007 |
0.006 |
0.009 |
0.011 |
0.010 |
0.010 |
0.009 |
0.011 |
0.012 |
0.006 |
0.008 |
0.006 |
0.007 |
0.009 |
0.010 |
0.012 |
0.009 |
0.012 |
0.009 |
0.011 |
0.010 |
0.007 |
0.016 |
0.010 |
0.005 |
0.012 |
0.010 |
0.009 |
0.012 |
0.008 |
0.008 |
0.009 |
0.009 |
| U |
— |
— |
— |
— |
0.001 |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
0.001 |
0.001 |
0.002 |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
— |
0.001 |
— |
— |
| Th |
— |
0.001 |
0.001 |
— |
0.001 |
0.001 |
— |
0.001 |
0.002 |
0.002 |
— |
— |
0.001 |
— |
— |
— |
0.012 |
0.009 |
0.007 |
— |
0.001 |
— |
— |
0.001 |
— |
— |
0.001 |
— |
— |
0.001 |
— |
0.001 |
— |
— |
— |
— |
0.001 |
0.002 |
— |
— |
| Al |
0.037 |
0.038 |
0.044 |
0.076 |
0.042 |
0.035 |
0.060 |
0.119 |
0.036 |
0.029 |
0.033 |
0.053 |
0.068 |
0.043 |
0.106 |
0.033 |
0.049 |
0.033 |
0.053 |
0.056 |
0.042 |
0.039 |
0.033 |
0.041 |
0.025 |
0.050 |
0.027 |
0.028 |
0.041 |
0.061 |
0.067 |
0.067 |
0.025 |
0.038 |
0.029 |
0.036 |
0.034 |
0.029 |
0.046 |
0.040 |
| Y |
0.010 |
0.013 |
0.013 |
0.014 |
0.013 |
0.021 |
0.010 |
0.017 |
0.010 |
0.008 |
0.011 |
0.009 |
0.006 |
0.010 |
0.005 |
0.009 |
0.037 |
0.025 |
0.021 |
0.018 |
0.020 |
0.008 |
0.009 |
0.017 |
0.012 |
0.019 |
0.011 |
0.013 |
0.010 |
0.002 |
0.010 |
0.004 |
0.011 |
0.014 |
0.014 |
0.009 |
0.014 |
0.012 |
0.010 |
0.005 |
| Fe |
0.013 |
0.015 |
0.023 |
0.024 |
0.015 |
0.015 |
0.032 |
0.025 |
0.019 |
0.016 |
0.011 |
0.010 |
0.103 |
0.015 |
0.011 |
0.015 |
0.022 |
0.026 |
0.022 |
0.013 |
0.029 |
0.026 |
0.018 |
0.031 |
0.015 |
0.012 |
0.015 |
0.017 |
0.012 |
0.031 |
0.014 |
0.011 |
0.014 |
0.016 |
0.019 |
0.017 |
0.016 |
0.010 |
0.011 |
0.017 |
| Ca |
0.030 |
0.029 |
0.031 |
0.021 |
0.023 |
0.019 |
0.025 |
0.033 |
0.036 |
0.028 |
0.015 |
0.021 |
0.023 |
0.026 |
0.021 |
0.021 |
0.039 |
0.031 |
0.038 |
0.020 |
0.017 |
0.017 |
0.020 |
0.014 |
0.019 |
0.024 |
0.023 |
0.026 |
0.032 |
0.027 |
0.020 |
0.022 |
0.023 |
0.030 |
0.027 |
0.016 |
0.030 |
0.025 |
0.030 |
0.031 |
| Pb |
— |
— |
0.001 |
— |
0.001 |
— |
— |
0.004 |
0.003 |
0.002 |
— |
— |
0.000 |
— |
— |
0.004 |
0.007 |
0.005 |
0.004 |
— |
0.001 |
— |
— |
— |
— |
— |
— |
— |
— |
— |
0.001 |
— |
— |
— |
— |
0.001 |
0.002 |
0.002 |
0.003 |
— |
| K |
0.002 |
0.001 |
0.002 |
0.003 |
0.001 |
— |
0.002 |
— |
— |
0.001 |
0.001 |
0.003 |
0.001 |
0.002 |
0.001 |
0.001 |
0.001 |
0.001 |
0.001 |
0.003 |
0.003 |
0.001 |
0.002 |
0.002 |
0.001 |
0.001 |
— |
0.002 |
0.001 |
— |
0.001 |
0.001 |
0.002 |
0.002 |
0.001 |
0.002 |
0.001 |
0.002 |
0.001 |
0.001 |
| Na |
0.004 |
0.009 |
0.006 |
0.016 |
0.009 |
0.006 |
0.017 |
0.036 |
0.005 |
0.011 |
0.011 |
0.016 |
0.051 |
0.027 |
0.049 |
0.009 |
0.003 |
0.010 |
0.005 |
0.013 |
0.015 |
0.032 |
0.008 |
0.010 |
0.009 |
0.009 |
0.005 |
0.010 |
0.004 |
0.020 |
0.039 |
0.037 |
0.004 |
0.014 |
0.054 |
0.013 |
0.005 |
0.008 |
0.006 |
0.007 |
| Sum |
2.038 |
2.043 |
2.047 |
2.059 |
2.040 |
2.035 |
2.060 |
2.090 |
2.043 |
2.040 |
2.033 |
2.044 |
2.120 |
2.056 |
2.079 |
2.037 |
2.059 |
2.053 |
2.055 |
2.047 |
2.052 |
2.058 |
2.037 |
2.046 |
2.034 |
2.042 |
2.031 |
2.041 |
2.037 |
2.058 |
2.067 |
2.062 |
2.031 |
2.049 |
2.073 |
2.038 |
2.041 |
2.036 |
2.040 |
2.041 |
|
附表 3 赛马岩体异霞正长岩异性石蚀变组合中锆石电子探针成分(wt%)
Appendix Table 3 Electron-microprobe results for zircon in different alteration associations from the lujavrite of the Saima complex (wt%)
|
表 5
Table 5
表 5(Table 5)

表 5
赛马岩体异霞正长岩异性石蚀变组合中钠沸石和钾长石电子探针成分(wt%)
Table 5
Electron-microprobe results for natrolite and K-feldspar in different alteration associations from the lujavrite of the Saima complex (wt%)
| 矿物 |
|
钠沸石 |
|
钾长石 |
| 序号 | |
1 |
2 |
3 |
4 |
5 | |
6 |
7 |
8 |
9 |
| SiO2 | |
47.28 |
47.15 |
46.24 |
46.20 |
47.24 | |
65.13 |
63.87 |
65.45 |
65.96 |
| TiO2 | |
0.00 |
— |
— |
— |
0.01 | |
— |
0.02 |
— |
— |
| ZrO2 | |
0.04 |
— |
— |
0.04 |
0.03 | |
— |
0.02 |
0.09 |
— |
| HfO2 | |
— |
0.07 |
0.03 |
— |
— | |
0.00 |
0.11 |
— |
0.02 |
| Al2O3 | |
25.86 |
27.43 |
27.98 |
26.07 |
26.69 | |
18.34 |
18.81 |
18.58 |
18.24 |
| FeO | |
0.05 |
0.01 |
0.46 |
0.02 |
0.25 | |
0.21 |
0.29 |
0.11 |
0.26 |
| CaO | |
0.55 |
0.15 |
0.42 |
0.84 |
1.58 | |
— |
0.04 |
0.02 |
0.00 |
| MgO | |
— |
— |
— |
— |
— | |
— |
— |
0.00 |
— |
| MnO | |
— |
— |
— |
— |
— | |
0.03 |
0.05 |
0.02 |
0.01 |
| K2O | |
0.15 |
0.09 |
0.08 |
0.05 |
0.05 | |
15.69 |
15.74 |
15.85 |
15.62 |
| Na2O | |
16.66 |
15.83 |
15.93 |
16.12 |
14.68 | |
0.09 |
0.14 |
0.10 |
0.12 |
| Total | |
90.59 |
90.73 |
91.15 |
89.34 |
90.13 | |
99.50 |
99.09 |
100.21 |
100.24 |
| O |
|
以10个氧原子计算 |
|
以8个氧原子计算 |
| Si | |
3.008 |
2.976 |
2.921 |
2.960 |
2.999 | |
3.012 |
2.977 |
3.006 |
3.024 |
| Ti | |
— |
— |
— |
— |
0.001 | |
— |
0.001 |
— |
— |
| Zr | |
0.001 |
— |
— |
0.001 |
0.001 | |
— |
— |
0.002 |
— |
| Hf | |
— |
0.001 |
0.001 |
— |
— | |
— |
0.001 |
— |
— |
| Al | |
1.939 |
2.041 |
2.083 |
1.969 |
1.997 | |
0.999 |
1.033 |
1.006 |
0.986 |
| Fe | |
0.003 |
0.001 |
0.024 |
0.001 |
0.013 | |
0.008 |
0.011 |
0.004 |
0.010 |
| Ca | |
0.037 |
0.010 |
0.028 |
0.058 |
0.108 | |
— |
0.002 |
0.001 |
— |
| Mg | |
— |
— |
— |
— |
— | |
— |
— |
— |
— |
| Mn | |
— |
— |
— |
— |
— | |
0.001 |
0.002 |
0.001 |
— |
| K | |
0.012 |
0.007 |
0.007 |
0.004 |
0.004 | |
0.926 |
0.936 |
0.929 |
0.913 |
| Na | |
2.054 |
1.937 |
1.952 |
2.027 |
1.858 | |
0.008 |
0.013 |
0.009 |
0.011 |
| Sum | |
7.054 |
6.973 |
7.016 |
7.020 |
6.981 | |
4.954 |
4.976 |
4.958 |
4.944 |
|
表 5 赛马岩体异霞正长岩异性石蚀变组合中钠沸石和钾长石电子探针成分(wt%)
Table 5 Electron-microprobe results for natrolite and K-feldspar in different alteration associations from the lujavrite of the Saima complex (wt%)
|
4.2.3 异性石假晶 部分样品中,霞石几乎完全被钠沸石交代,与霞石共生的异性石发生严重蚀变,形成了假晶单颗粒或集合体(图 4e, f)。进一步观察发现,异性石假晶主要由残余异性石+钠锆石+锆石+钠沸石+霓石+钾长石+铈硅磷灰石组成(图 4g-i、图 6)。残余异性石一般位于假晶内部,周围被钠锆石等次生矿物交代(图 4i)。钠锆石呈片状,宽度 < 10μm,多呈放射状集合体形式出现;霓石呈针状、纤维状,长度 < 20μm,宽度 < 5μm,也多以放射状集合体与钠锆石共生;铈硅磷灰石是假晶中主要的稀土矿物,呈粒状(粒径 < 10μm),与锆石一起零星地分布于钠沸石周围(图 4g),部分铈硅磷灰石颗粒“逃逸”出异性石假晶,呈粒状或细脉状分布于周围的钠沸石中(图 4h, i)。假晶中钾长石不同于造岩矿物,多呈他形,并具有较多的孔洞,包裹钠锆石、铈硅磷灰石等矿物(图 4h);少量钾长石呈细小颗粒集合体(粒径 < 10μm)分布于假晶周围的钠沸石中(图 4i)。
与之前蚀变组合相似,假晶中钠锆石总量89.98%~92.63%,其中含有3.12%~4.07% CaO和10.14%~12.03% Na2O(表 2);锆石总量为95.24%~98.47%,其中Al2O3、FeOT和CaO含量分别为0.67%~1.49%、0.48%~1.16%和0.42%~0.60%(表 4、附表 3、图 5)。相比于作为造岩矿物的霓石(Wu et al., 2016, 0.19%~1.74% ZrO2,-0.05% Nb2O5,表 3、附表 2),假晶中次生霓石含有较高的Zr(3.03%~5.96% ZrO2)和Nb(0.16%~1.06% Nb2O5)含量。假晶中部分钾长石除含有主量元素K(15.61%~15.85% K2O)、Al(18.24%~18.80% Al2O3)、Si(63.87%~65.96% SiO2)外,部分颗粒ZrO2含量可达0.09%(检测限200×10-6,表 5)。铈硅磷灰石作为假晶中主要的轻稀土矿物,主要含有20.42%~25.74% SiO2、57.74%~66.87% REE2O3(其中主要为9.83%~16.81% La2O3、22.38%~34.51% Ce2O3和8.26%~11.85% Nd2O3),3.71%~12.42% CaO和0.93%~2.53% F(表 6)。
表 6
Table 6
表 6(Table 6)

表 6
赛马岩体异霞正长岩异性石假晶中铈硅磷灰石电子探针成分(wt%)
Table 6
Electron-microprobe results for britholite-(Ce) in the pseudomorph after eudialyte from the lujavrite of the Saima complex (wt%)
| 矿物 |
|
异性石假晶中铈硅磷灰石 |
| 序号 | |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
10 |
11 |
12 |
| P2O5 | |
0.18 |
0.14 |
0.03 |
— |
0.04 |
1.88 |
0.23 |
1.21 |
1.10 |
0.04 |
— |
— |
| Nb2O5 | |
— |
0.02 |
0.14 |
0.11 |
0.04 |
0.08 |
0.17 |
0.13 |
0.04 |
0.01 |
— |
— |
| SiO2 | |
21.46 |
22.49 |
24.47 |
25.74 |
23.68 |
20.42 |
25.14 |
20.88 |
22.12 |
25.03 |
21.08 |
20.69 |
| TiO2 | |
0.01 |
0.05 |
0.67 |
0.70 |
0.37 |
0.04 |
0.85 |
0.68 |
0.16 |
0.17 |
— |
— |
| ZrO2 | |
— |
0.23 |
0.14 |
0.18 |
0.31 |
0.07 |
0.52 |
0.29 |
0.09 |
— |
— |
0.06 |
| HfO2 | |
0.08 |
0.01 |
— |
— |
0.01 |
0.07 |
0.14 |
0.09 |
0.02 |
— |
0.03 |
0.19 |
| ThO2 | |
0.56 |
0.55 |
0.05 |
— |
0.45 |
1.96 |
1.76 |
5.90 |
3.55 |
0.07 |
0.73 |
1.23 |
| UO2 | |
0.01 |
— |
— |
0.04 |
— |
— |
0.01 |
0.07 |
0.05 |
— |
0.09 |
0.10 |
| Al2O3 | |
— |
— |
0.44 |
0.44 |
0.03 |
0.07 |
0.63 |
0.70 |
0.20 |
0.46 |
— |
0.01 |
| La2O3 | |
15.27 |
13.45 |
12.43 |
12.53 |
11.99 |
9.83 |
12.00 |
10.70 |
10.70 |
18.42 |
15.43 |
14.05 |
| Ce2O3 | |
32.52 |
32.57 |
29.29 |
29.26 |
31.21 |
24.69 |
23.35 |
22.38 |
24.34 |
26.20 |
32.51 |
30.56 |
| Pr2O3 | |
1.89 |
1.97 |
1.97 |
2.05 |
2.17 |
2.15 |
2.00 |
2.00 |
2.03 |
2.49 |
3.66 |
3.62 |
| Nd2O3 | |
8.55 |
8.26 |
10.27 |
10.69 |
11.43 |
13.30 |
10.19 |
11.31 |
11.85 |
10.13 |
11.78 |
11.32 |
| Sm2O3 | |
1.15 |
1.67 |
5.09 |
1.23 |
1.76 |
4.99 |
2.54 |
4.52 |
2.63 |
1.41 |
1.17 |
1.14 |
| Eu2O3 | |
0.88 |
0.86 |
0.92 |
0.82 |
1.08 |
1.43 |
1.31 |
1.32 |
1.46 |
0.97 |
0.07 |
0.22 |
| Gd2O3 | |
4.68 |
4.52 |
3.33 |
3.78 |
4.23 |
4.22 |
4.57 |
3.96 |
4.43 |
3.83 |
0.59 |
0.79 |
| Tb2O3 | |
0.06 |
— |
— |
— |
— |
— |
0.02 |
— |
— |
— |
— |
— |
| Dy2O3 | |
0.17 |
0.42 |
0.25 |
0.08 |
— |
0.04 |
0.42 |
0.42 |
0.39 |
0.43 |
— |
— |
| Ho2O3 | |
0.46 |
0.53 |
— |
— |
0.02 |
0.23 |
0.98 |
0.45 |
0.73 |
— |
0.12 |
0.07 |
| Er2O3 | |
0.01 |
0.05 |
0.08 |
0.05 |
0.07 |
— |
0.02 |
0.04 |
0.02 |
0.01 |
— |
— |
| Tm2O3 | |
0.04 |
0.03 |
— |
— |
— |
0.05 |
0.10 |
0.11 |
0.13 |
0.04 |
0.15 |
— |
| Yb2O3 | |
— |
— |
— |
— |
0.02 |
— |
0.00 |
— |
— |
— |
— |
— |
| Y2O3 | |
0.13 |
0.16 |
0.03 |
0.04 |
0.02 |
0.17 |
0.41 |
0.55 |
0.41 |
— |
0.29 |
0.32 |
| CaO | |
10.02 |
9.87 |
6.96 |
7.38 |
5.63 |
12.42 |
7.14 |
10.39 |
10.16 |
6.79 |
9.10 |
10.39 |
| MgO | |
— |
— |
0.01 |
0.01 |
— |
— |
0.02 |
0.01 |
— |
0.02 |
— |
— |
| FeO | |
0.49 |
0.72 |
2.60 |
2.68 |
3.37 |
0.14 |
3.23 |
0.83 |
1.12 |
2.05 |
0.74 |
0.85 |
| MnO | |
— |
— |
0.97 |
1.02 |
0.14 |
— |
0.28 |
0.15 |
0.04 |
1.71 |
— |
— |
| SrO | |
0.06 |
0.03 |
0.30 |
0.25 |
0.38 |
0.52 |
0.40 |
0.51 |
0.34 |
0.52 |
1.86 |
2.99 |
| BaO | |
0.06 |
— |
0.11 |
0.07 |
0.01 |
0.04 |
0.03 |
0.05 |
0.03 |
— |
— |
— |
| PbO | |
0.20 |
0.11 |
0.05 |
— |
0.03 |
0.09 |
0.08 |
0.14 |
0.17 |
— |
— |
— |
| Na2O | |
— |
0.15 |
0.19 |
0.24 |
0.23 |
0.02 |
0.04 |
— |
0.17 |
0.01 |
— |
0.12 |
| K2O | |
0.02 |
— |
0.01 |
0.02 |
0.03 |
— |
0.01 |
0.02 |
0.01 |
0.03 |
0.03 |
0.03 |
| F | |
2.03 |
2.15 |
1.45 |
1.63 |
1.55 |
1.96 |
1.17 |
0.93 |
1.35 |
1.40 |
2.53 |
2.72 |
| Cl | |
0.06 |
0.09 |
0.07 |
0.08 |
0.08 |
0.08 |
0.09 |
0.10 |
0.10 |
0.03 |
— |
— |
| F=O | |
0.93 |
0.99 |
0.67 |
0.75 |
0.71 |
0.90 |
0.54 |
0.43 |
0.62 |
0.64 |
1.17 |
1.25 |
| Cl=O | |
0.01 |
0.02 |
0.02 |
0.02 |
0.02 |
0.02 |
0.02 |
0.02 |
0.02 |
0.01 |
— |
— |
| Total | |
100.09 |
100.07 |
101.63 |
100.32 |
99.62 |
100.03 |
99.30 |
100.37 |
99.29 |
101.61 |
100.80 |
100.18 |
| 根据∑(P+Si+Zr)=6阳离子计算 |
| P | |
0.043 |
0.031 |
0.005 |
— |
0.008 |
0.434 |
0.046 |
0.279 |
0.242 |
0.008 |
— |
— |
| Nb | |
— |
0.002 |
0.015 |
0.012 |
0.005 |
0.010 |
0.018 |
0.016 |
0.005 |
0.001 |
— |
— |
| Si | |
5.957 |
5.940 |
5.979 |
5.980 |
5.954 |
5.557 |
5.893 |
5.683 |
5.747 |
5.992 |
6.000 |
5.992 |
| Ti | |
0.001 |
0.012 |
0.123 |
0.123 |
0.069 |
0.008 |
0.150 |
0.140 |
0.031 |
0.030 |
— |
— |
| Zr | |
— |
0.029 |
0.016 |
0.020 |
0.038 |
0.009 |
0.060 |
0.038 |
0.011 |
0.000 |
— |
0.008 |
| Hf | |
0.007 |
— |
— |
— |
0.001 |
0.005 |
0.010 |
0.007 |
0.001 |
— |
0.002 |
0.016 |
| Th | |
0.035 |
0.033 |
0.002 |
— |
0.026 |
0.121 |
0.093 |
0.366 |
0.211 |
0.004 |
0.047 |
0.081 |
| U | |
0.001 |
— |
— |
0.002 |
— |
— |
0.001 |
0.005 |
0.003 |
— |
0.006 |
0.006 |
| Al | |
— |
— |
0.126 |
0.121 |
0.009 |
0.024 |
0.175 |
0.226 |
0.059 |
0.129 |
— |
0.002 |
| La | |
1.563 |
1.311 |
1.121 |
1.073 |
1.113 |
0.988 |
1.039 |
1.074 |
1.025 |
1.627 |
1.617 |
1.498 |
| Ce | |
3.306 |
3.150 |
2.620 |
2.489 |
2.874 |
2.460 |
2.004 |
2.231 |
2.315 |
2.296 |
3.386 |
3.238 |
| Pr | |
0.191 |
0.199 |
0.175 |
0.173 |
0.199 |
0.213 |
0.171 |
0.198 |
0.192 |
0.217 |
0.379 |
0.381 |
| Nd | |
0.848 |
0.745 |
0.896 |
0.888 |
1.026 |
1.293 |
0.853 |
1.100 |
1.099 |
0.866 |
1.197 |
1.170 |
| Sm | |
0.110 |
0.146 |
0.429 |
0.099 |
0.153 |
0.468 |
0.205 |
0.424 |
0.234 |
0.116 |
0.115 |
0.113 |
| Eu | |
0.083 |
0.077 |
0.077 |
0.065 |
0.092 |
0.133 |
0.104 |
0.122 |
0.129 |
0.079 |
0.007 |
0.022 |
| Gd | |
0.430 |
0.396 |
0.269 |
0.291 |
0.353 |
0.380 |
0.355 |
0.357 |
0.382 |
0.304 |
0.055 |
0.076 |
| Tb | |
0.006 |
— |
— |
— |
— |
— |
0.001 |
— |
— |
— |
— |
— |
| Dy | |
0.014 |
0.035 |
0.019 |
0.006 |
— |
0.003 |
0.031 |
0.038 |
0.033 |
0.033 |
— |
— |
| Ho | |
0.040 |
0.044 |
— |
— |
0.002 |
0.021 |
0.073 |
0.039 |
0.060 |
— |
0.011 |
0.006 |
| Er | |
0.001 |
0.003 |
0.006 |
0.003 |
0.005 |
— |
0.002 |
0.003 |
0.001 |
0.001 |
— |
— |
| Tm | |
0.003 |
0.002 |
— |
— |
— |
0.005 |
0.008 |
0.009 |
0.011 |
0.003 |
0.014 |
— |
| Yb | |
— |
— |
— |
— |
0.002 |
— |
— |
— |
— |
— |
— |
— |
| Y | |
0.019 |
0.022 |
0.004 |
0.005 |
0.003 |
0.025 |
0.051 |
0.079 |
0.057 |
— |
0.043 |
0.050 |
| Ca | |
2.982 |
2.794 |
1.823 |
1.836 |
1.517 |
3.621 |
1.794 |
3.030 |
2.829 |
1.742 |
2.774 |
3.224 |
| Mg | |
— |
— |
0.005 |
0.002 |
— |
— |
0.008 |
0.002 |
— |
0.006 |
— |
— |
| Fe | |
0.114 |
0.143 |
0.531 |
0.521 |
0.708 |
0.032 |
0.633 |
0.188 |
0.243 |
0.410 |
0.176 |
0.204 |
| Mn | |
— |
— |
0.201 |
0.200 |
0.031 |
— |
0.055 |
0.035 |
0.008 |
0.346 |
— |
— |
| Sr | |
0.009 |
0.004 |
0.042 |
0.033 |
0.055 |
0.082 |
0.054 |
0.081 |
0.052 |
0.073 |
0.307 |
0.502 |
| Ba | |
0.007 |
— |
0.010 |
0.006 |
0.001 |
0.005 |
0.003 |
0.005 |
0.003 |
— |
— |
— |
| Pb | |
0.014 |
0.008 |
0.003 |
— |
0.002 |
0.007 |
0.005 |
0.010 |
0.012 |
— |
— |
— |
| Na | |
— |
0.077 |
0.092 |
0.108 |
0.111 |
0.009 |
0.018 |
— |
0.085 |
0.006 |
— |
0.065 |
| K | |
0.008 |
0.001 |
0.002 |
0.005 |
0.008 |
— |
0.004 |
0.008 |
0.002 |
0.009 |
0.012 |
0.010 |
| F | |
1.775 |
1.792 |
1.120 |
1.192 |
1.232 |
1.681 |
0.864 |
0.798 |
1.108 |
1.055 |
2.277 |
2.489 |
| Cl | |
0.029 |
0.042 |
0.030 |
0.032 |
0.033 |
0.037 |
0.036 |
0.046 |
0.045 |
0.014 |
— |
— |
|
表 6 赛马岩体异霞正长岩异性石假晶中铈硅磷灰石电子探针成分(wt%)
Table 6 Electron-microprobe results for britholite-(Ce) in the pseudomorph after eudialyte from the lujavrite of the Saima complex (wt%)
|
5 讨论 5.1 异性石成分变化与碱性岩浆演化的关系 异性石是碱性岩的特征副矿物之一,广泛分布于俄罗斯科拉半岛Lovozero、Khibina、南格陵兰Ilímaussaq、南非Pilansberg等碱性杂岩体中(Mitchell and Chakhmouradian, 1996, 1998; Olivo and Williams-Jones, 1999; Karup-Møller et al., 2010; Marks and Markl, 2015)。前人研究表明异性石化学成分与结晶环境具有密切的关系(Schilling et al., 2011; Chakrabarty et al., 2012; Mitchell and Chakrabarty, 2012)。赛马异霞正长岩中多期异性石,从早到晚经历了Mn、Nb、REE含量上升,Si、Fe、Cl含量下降,Sr含量先上升后下降的变化过程(表 1、附表 1、图 3),为研究该地区碱性岩浆演化过程中体系成分变化和元素分异特点提供重要信息。
异性石Mn/Fe比值主要受(1)结晶时熔体Fe、Mn含量;(2)Fe、Mn在异性石-熔体间分配系数(受温度和熔体成分控制);(3)共生Fe、Mn矿物共同影响(Schilling et al., 2011)。前人研究表明,异性石Mn/Fe比值可作为判断碱性岩浆分异演化程度的指标之一,一般来说演化程度较高的碱性熔体中结晶出的异性石具有较高的Mn/Fe比值(Johnsen and Gault, 1997; Pfaff et al., 2008; Schilling et al., 2009, 2011)。从早期到晚期,赛马异霞正长岩异性石Mn/Fe比值依次上升,与前人研究结果一致,也佐证了我们观察判断的正确性。据Wu et al., 2016, 赛马异霞正长岩中主要的含Fe矿物为霓石(占造岩矿物的30%~40%),而无明显Mn的载体矿物,早期霓石的结晶是导致残余熔体和异性石Mn/Fe比值上升的主要原因。异性石中Si含量不断降低可能与熔体硅不饱和程度有密切关系,赛马碱性杂岩区经历了由硅饱和正长岩(石英正长岩、粗面岩)向硅不饱和正长岩(响岩、霞石正长岩和异霞正长岩)的演化(Zhu et al., 2016),异霞正长岩形成于赛马碱性杂岩区晚阶段,该岩性具有最低的全岩SiO2含量(约51%~52%, Wu et al., 2016; Zhu et al., 2016),随着异霞正长岩过碱质硅酸盐熔体钾长石、霞石和霓石等造岩矿物的早期结晶,残余熔体碱质不断降低而硅不饱和程度越发强烈,从而导致了异性石Si含量不断降低的结果。异霞正长岩异性石中Nb和REE含量不断增加与其不相容元素自身性质有关,这些不相容元素很难进入早期结晶的矿物晶体结构中,而在残余岩浆中相对富集,随着岩浆演化和温度降低,最终进入晚期异性石中(图 7a, b);此外,REE在异性石/硅酸盐熔体的分配系数随着熔体碱质的降低而增大(Marks et al., 2008),也可能是造成晚期异性石富REE的原因之一。Sr作为不相容元素,变化趋势本应与Nb、REE相似,但异性石中Sr含量经历了先上升后下降的变化过程,可能与其他共生的富Sr矿物(如大量闪叶石[(Na, Mn, K)3(Sr, Ba)2Ti3O2(O, OH, F)2])的结晶有关(Wu et al., 2016),随着大量的Sr进入到这些矿物中,导致相对较少的Sr进入到晚期异性石中。晚期异性石较低的Cl含量,可能与早期异性石的结晶消耗了熔体中的Cl或Cl作为挥发分逃逸有关。
5.2 异性石蚀变组合与多期次热液流体作用的关系 5.2.1 阶段1:钠交代——钠锆石的形成 异性石是碱性岩中相对容易受流体改造的副矿物,类似的异性石热液蚀变广泛发生于南格陵兰Ilímaussaq、North Qôroq、南非Pilansberg和印度Sushina等地,但蚀变产物不尽相同(Coulson, 1997; Mitchell and Liferovich, 2006; Karup-Møller et al., 2010; Mitchell and Chakrabarty, 2012; Karup-Møller and Rose-Hansen, 2013)。赛马异霞正长岩中,被霓石包裹的异性石经历了早期的热液交代,形成了异性石+钠锆石+霓石±钠沸石等富钠次生矿物(图 4a、图 7c),属于碱性岩晚期典型的钠交代过程。
前人研究表明,钠锆石等富钠次生矿物的形成与流体H2O活动性增加、体系温度降低有着密切的联系(Chakhmouradian and Mitchell, 2002; Andersen et al., 2010, 2014)。考虑到异霞正长岩熔体本身富钠的性质(Wu et al., 2016),且缺少来自围岩中钠交代的相关报道,引起异性石蚀变形成钠锆石+霓石±钠沸石的流体很可能来自于碱性熔体晚期形成的富Na、F、H2O的自交代流体(Johnsen and Grice, 1999; Mitchell and Liferovich, 2006; Chakrabarty et al., 2012)。次生矿物组合中Zr、Fe主要来自于异性石,而Na、Al、Si等元素则可能部分来自于交代流体或残余熔体。由于流体作用初期较弱的水-岩相互作用且周围霓石不易受流体影响的性质,导致这种轻微的蚀变类型多发生于异性石裂隙或边缘。
5.2.2 阶段2:强烈碱交代——锆石的形成 不同于阶段1,部分与钾长石和霞石共生的异性石进一步遭受了更加强烈的热液蚀变,形成了异性石+钠锆石+锆石+钠沸石±霓石的蚀变组合(图 4c, d、图 7d)。由于钠锆石常作为原生锆矿物的次生蚀变矿物而存在,不论从实验相图还是野外实例,尚无次生钠锆石在热液流体作用下进一步蚀变成锆石的报道(Chakhmouradian and Mitchell, 2002; Andersen et al., 2010),且薄片中未观察到明显的富锆流体作用,因此我们认为次生锆石直接来自于异性石的蚀变。相似地,异性石蚀变成锆石的现象也存在于摩洛哥Tamazeght、南非Pilansberg、加拿大Thor Lake和南格陵兰Ilímaussaq碱性岩体中(Salvi et al., 2000; Mitchell and Liferovich, 2006; Karup-Møller et al., 2010; Sheard et al., 2012),而这一过程可以用异性石的溶解-再沉淀模型来解释。
异霞正长岩晚期富Na、F的自交代流体使得部分异性石发生溶解蚀变,Zr等高场强元素与F形成复杂络合物的形式发生迁移(Migdisov et al., 2011; Migdisov and Williams-Jones, 2014),由于周围造岩矿物矿物早已结晶,留给这些络合物的迁移空间有限(图 4c)。当络合物遇到另一种性质的流体(可能富Ca)时,络合物的稳定性遭到破坏,从而再沉淀形成锆石等次生矿物。与赛马霞石正长岩钾锆石蚀变组合中次生锆石进行对比(Wu et al., 2015, 图 5),异性石蚀变组合中次生锆石具有更高的Al(0.67%~1.49% Al2O3)含量和更低的Ca(0.42%~0.60% CaO)含量;与其他异性石蚀变组合对比(Mitchell and Liferovich, 2006; Karup-Møller et al., 2010; Estrade et al., 2014),赛马异性石蚀变组合中未出现大量次生锆石和方解石、萤石等能够指示强烈富Ca流体的矿物,因此我们认为蚀变过程中富Na、Al、F的自交代流体占主导而仅存在少量引起锆石再沉淀的富Ca流体。考虑到异霞正长岩周围并无太多钙质围岩(图 1),而且异霞正长岩全岩Sr-Nd-Hf同位素信息与主体霞石正长岩基本一致且变化不大(Zhu et al., 2016),少量的富Ca流体可能是由于早期富Na、Al和F的流体交代早期异性石,从异性石中迁移出的Ca所致。
5.2.3 阶段3:多期热液流体叠加作用——假晶的形成 与霞石共生的异性石经历了一系列强烈的热液蚀变,形成了由残余异性石+钠锆石+锆石+钠沸石+霓石+钾长石+铈硅磷灰石组成的假晶,与此同时周围的霞石也几乎完全被钠沸石交代(图 4f-i、图 7e)。与阶段2中蚀变组合相比,假晶中新增了次生钾长石和铈硅磷灰石,值得我们去进一步讨论。
区别于造岩矿物,假晶中钾长石较多孔洞的特点(图 4h)及其假晶周围钾长石细小颗粒(图 4i)暗示了其很可能来源于热液流体而不是碱性岩浆。但在之前的异性石蚀变组合中未发现钾长石及其他含钾次生矿物,原因可能是:(1)富K流体是区别于富Na、Al、F的自交代流体的另一股流体,并晚于自交代流体交代异性石从而形成次生钾长石;(2)即使自交代流体中含K,但在较低的水/岩比例下,K不同于Na,在碱性流体中较难与Fe3+、Zr结合形成稳定的次生矿物(如钠锆石、霓石)而沉积(Markl et al., 2010),而在流体作用晚期,水/岩比例增加、温度降低的条件下,最终形成钾长石。
铈硅磷灰石是假晶中主要的富REE、Ca和F的次生矿物,与锆石的溶解-再沉淀过程相似,铈硅磷灰石的形成是富F、H2O的自交代流体与富Ca流体叠加作用的结果,其中REE主要来自于异性石。值得注意的是,相比于锆石,部分铈硅磷灰石已经迁移出异性石假晶而出现在周围钠沸石中(图 4h)产生的原因包括:(1)REE比Zr具有更强的热液活动性,REE-F络合物在流体中易于迁移(Sheard et al., 2012);(2)假晶周围霞石同样遭受严重蚀变(形成钠沸石),为REE的运移提供了一定空间。
结合异性石化学成分及各次生矿物元素组成可知,异性石中绝大部分元素在次生矿物中得以保存,例如Zr主要赋存于钾锆石和锆石,少量赋存于霓石,Na、Fe主要赋存于霓石,Ca、REE主要赋存于铈硅磷灰石;然而各次生矿物中均不含Cl(或含量极低),可以推断出早期异性石的结晶消耗了熔体中的Cl,或Cl作为挥发分从体系中逃逸,这与晚期异性石Cl含量降低相吻合(表 1、附表 1)。同时,通过对异性石各蚀变组合进行对比,我们推断出其至少经历过两阶段的热液流体叠加相互作用:(1)占主导的富Na(±K)、Al、F的自交代流体;(2)少量富Ca流体,异性石完整的热液蚀变过程见图 7。
5.3 高场强元素的热液活动性对碱性岩成矿作用的指示意义 通过对异性石及其他稀有、稀土矿物研究,学者发现这些矿物在某些特殊的地质环境中并不稳定,在热液影响下Zr和REE等高场强元素易于与F、Cl等阴离子形成复杂的络合物从而增加此类高场强元素的热液活动性,使得其可以在热液中运移几微米至几十米不等(Salvi and Williams-Jones, 1996; Williams-Jones et al., 2012; Gysi and Williams-Jones, 2013; Li et al., 2015),对于研究热液流体与碱性岩成矿作用具有重要的意义。
对于热液成因或受热液流体影响的稀有、稀土矿床,流体在成矿过程中起到“搬运”和“提纯”的作用。一方面稀有、稀土元素与流体中F、Cl等阴离子形成复杂的络合物,随着热液流体运移,当温度降低或者有“沉淀剂”加入时形成有经济价值的稀有、稀土矿床;另一方面,原生的稀有、稀土矿物中稀有、稀土元素通过“溶解-再沉淀”过程,形成次生矿物的稀有、稀土元素含量远高于原生矿物,如赛马异霞正长岩异性石假晶中次生锆石的Zr含量、铈硅磷灰石的REE含量远高于原生异性石。当然,高场强元素的热液活动性与流体成分、盐度等性质、体系温度、pH等物理化学条件紧密相关(Wood, 1990; Migdisov et al., 2009, 2011; Migdisov and Williams-Jones, 2014),在研究某一具体的高场强元素的热液活动性时需要特别注意。
6 结论 (1) 辽宁赛马碱性岩体异霞正长岩中发育多期异性石。从早到晚,异性石经历了Mn、Nb、REE含量上升,Si、Fe、Cl含量下降,Sr含量先上升后下降的变化过程,与碱性岩浆演化、不相容元素自身性质、共生矿物结晶及挥发分逃逸有着密不可的联系。
(2) 异性石经历了一系列热液流体叠加相互作用,最终形成了由残余异性石+钠锆石+锆石+钠沸石+霓石+钾长石+铈硅磷灰石组成的假晶,其中锆石具有富Al、Fe、Ca的特点。
(3) 异性石假晶的形成可用矿物“溶解-再沉淀”模型进行解释,引起蚀变的流体至少包括:1)占主导的富Na(±K)、Al、F的自交代流体;2)少量富Ca流体。异性石在自交代流体影响下,首先经历钠交代,形成钠锆石等次生矿物,当水-岩相互作用进一步强烈使得异性石发生溶解,可能形成Zr-F、REE-F络合离子,当遇到富Ca流体时,络合离子以锆石、铈硅磷灰石的形式发生再沉淀,最终形成假晶。
致谢
野外工作获得了中国科学院地质与地球物理研究所杨进辉研究员、朱昱升博士、陈璟元博士的大力支持,室内电子探针分析工作获得了南京大学地球科学与工程学院张文兰教授级高级工程师的悉心帮助,在此深表谢意。同时,感谢王连训博士及另一名匿名审稿人对本文提供的宝贵建议和修改意见。
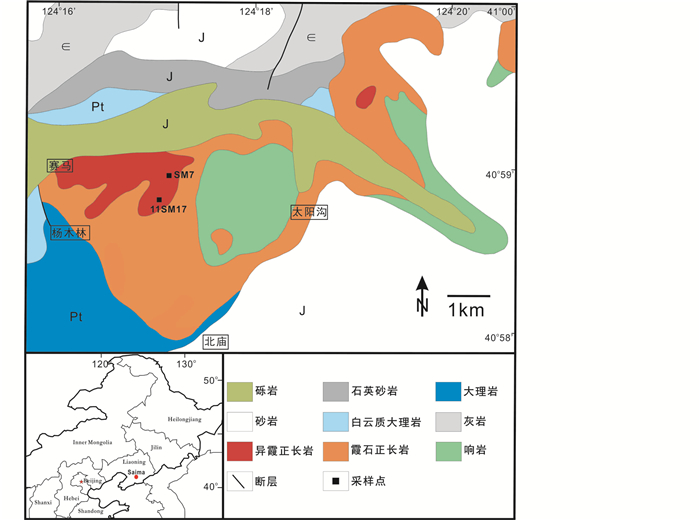

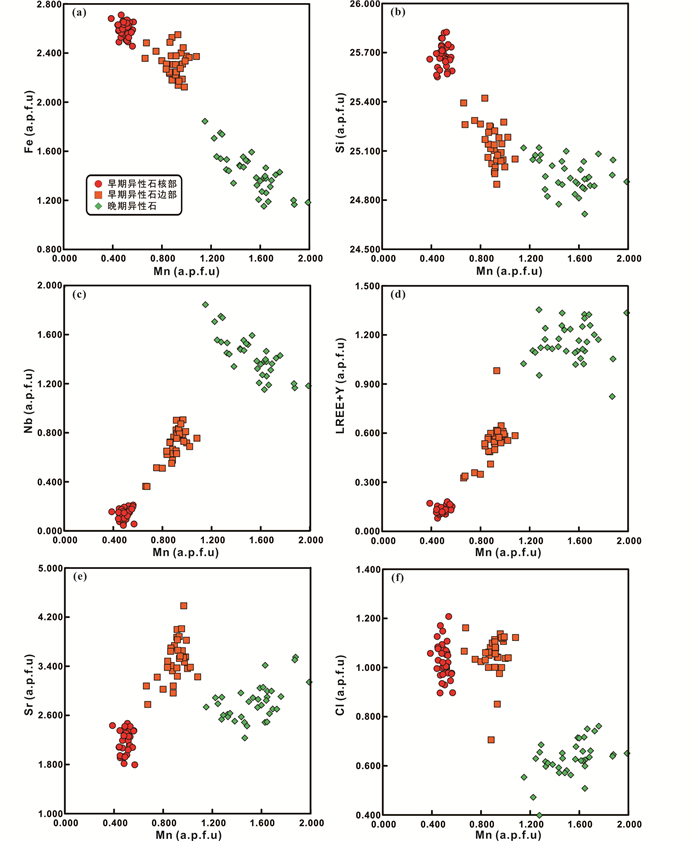

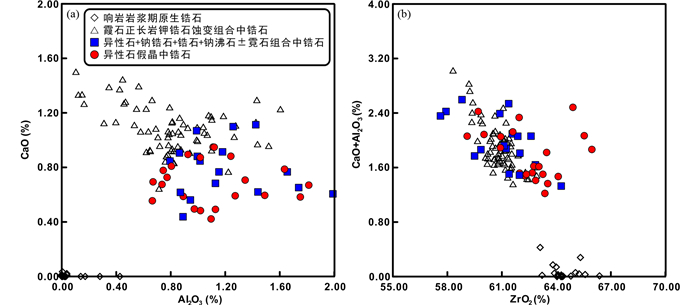

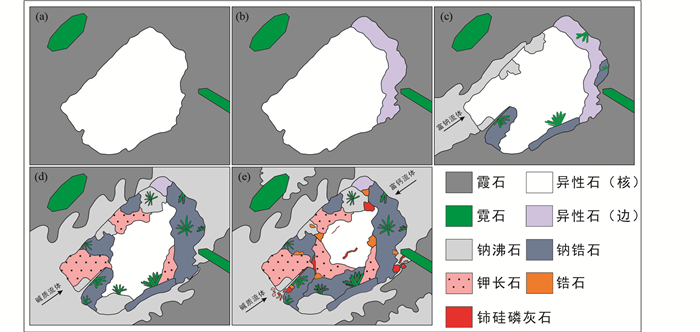
 2018, Vol. 34
2018, Vol. 34


