文章信息
- Yang Liujun, Ma Ling, Liu Xiaoyu, Feng Rui, Zhu Guilan, Zeng Aiping
- 杨柳君, 马玲, 刘晓玉, 冯睿, 朱桂兰, 曾爱平
- Sexual Selection, Mating and Fecundity of Basilepta melanopus (Coleoptera: Chrysomelidae), A Defoliator of Camellia Oleifera
- 油茶食叶害虫角胸叶甲性选择和交配、产卵等行为学
- Scientia Silvae Sinicae, 2018, 54(1): 74-80.
- 林业科学, 2018, 54(1): 74-80.
- DOI: 10.11707/j.1001-7488.20180108
-
文章历史
- Received date: 2017-03-02
- Revised date: 2017-05-08
-
作者相关文章
Basilepta melanopus(Coleoptera:Chrysomelidae) was reported in 1986 to be an important new pest of tea(Camellia sinensis)(Tan et al., 1986) and was later reported to also attack oil tea(Camellia oleifera), a commercial source of tea oil(He et al., 2011).This pest is now widely distributed in the tea growing provinces of China(Fujian, Guangdong, Hubei, Hunan, Guangxi, Jiangxi).In Guangxi Province, damage caused by this pest had become severe by 2005 in Shaoping County(Zhu, 2013), and there have also been recent outbreaks in Jiangxi Province(Wang et al., 2004).
B.melanopus has one generation per year in Hunan Province, where it overwinters as larvae in the soil, with adults emerging from late April onward.The adult flies to the host plant to eat the leaves, which become damaged by a large number of semi-transparent circular holes, which affect photosynthesis and production(Li et al., 2014).Rearing B.melanopus in the laboratory is difficult because the larvae live in the soil.So far, research has mostly focused on the biological characteristics and control of adults in China(Zeng et al., 2012; Jie et al., 2014; Li et al., 2013).However, the mating, oviposition, and sexual selection behaviors of this species have seldom been studied.Such information could lead to better methods for trapping of these beetles.based on use of their sex pheromones to trap or repel this pest.Also, better understanding of how to rear this species in the laboratory might incentivize more researchers to study the larvae and adults.
1 Materials and methods 1.1 Source of insects and feedingMature larvae were collected from the soil at Yong'an(Changsha, Hunan), an oil tea plantation, in early March 2016, and held individually in soil-filled tubes(5 mL) at outdoor temperature under L:D=14:8 until eclosion.After emergence, the adults were transferred into the transparent plastic boxes(14 cm×14 cm× 6 cm) individually and oil tea leaves were added to it as food for the adults.
1.2 Observations of matingOne male-female pair that had emerged on the same day were placed in a plastic box.Mating behavior was then continuously observed by human eyes.Copulation behavior, which consisted of pre-copulation, copulation, and post-copulation stages was observed for ten couples.
1.3 Circadian rhythm of mating and ovipositionTen pairs of adults that emerged on the same day, were placed into one plastic box to observe circadian rhythms of mating and oviposition by human eyes for three days.The test was replicated three times.
1.4 Egg stage, hatching rates and mortality of larvae under different temperature regimesEight female adults ready to oviposit were individually placed in plastic boxes and held at outdoor temperature to observe oviposition and to record the number of eggs laid.The resulting eggs were divided into three treatments and held in an incubator until all eggs hatched, at 23, 26, and 29 ℃, respectively.Hatching and mortality rates were recorded at each temperature.
1.5 Sexual selectionTo observe what effect male size had on mate selection by females, two male adults with a large difference in weight were marked, black for the large one(6.0-6.5 mg) and red for the small one(4.1-4.5 mg), and placed together with one similarly median aged female on a fresh oil tea branch in a plastic box.Mating behavior was observed for 10 days and the frequency of successful mating by males in each size class was determined.Each trio of beetles was replicated 10 times.
In a second experiment, two females with a large difference in weight were similarly marked, black for the large(6.1-7.3 mg) one and red for the small(4.2-5.0 mg) one, and both were placed together with one similarly median aged male on a fresh oil tea branch in a plastic box.Mating behavior was observed for 10 days and the difference in frequency of successful mating between the large and the small females was analyzed.Each trio of beetles was replicated 10 times.
1.6 Effect of mating experience on sexual selectionThis experiment had two variations, each replicated 10 times.All males and females used were of a similar size, and all replicates were held in plastic boxes as described above.In the first variation, one mated male and one virgin male(Y♂ & N♂) were placed together with a virgin female(N♀).In the second variation, a virgin male(N♂) was put together with a mated female and a virgin female(Y♀ & N♀).
1.7 Data analysisExcel 2007 and SPSS 21.0(Xu, 2001)were used to analyze the data.The Levene test and the Kolmogorov-Smirnov test were used to check for homoscedasticity and normality.Differences between treatments within experiments were compared using a least significant differential method test(LSD).
2 Result 2.1 Mating behavior of B.melanopusMating behavior can be divided into 3 stages:pre-copulation, copulation, and post-copulation(in chronological order).During pre-copulation, males mostly approached females from behind(68.7%) but occasionally the male may turn around after mounting the back of female from the front(20.3%) or via the side(11.0%) to make sure their heads are in the same direction.The male protrudes its aedeagus immediately after mounting the female's back, and the female vigorously moves her body from side to side.The male will re-mount the female if he is prematurely dislodged by the female, until the female stops struggling.Usually the female will not move(97.6%) during mating, but rarely(2.4%) the female goes on feeding while carrying the male.During post-copulation, most males(81.0%) remain on the female's back for a while before moving away.A small proportion(12.3%) of mating events end with the female leaving first and an even smaller portion(6.8%) with the male and female leaving together.
B.melanopus can mate and oviposit up to a dozen times in their lifetime, and can mate at any time during a day.Mating behavior lasted on average (18.9±9.3) min(mean±SD), and for most males the total duration was between 16 and 30 min(58.8% of all matings in this range, n=136;Fig. 1)(df=7, F=799.6, P=0.000).
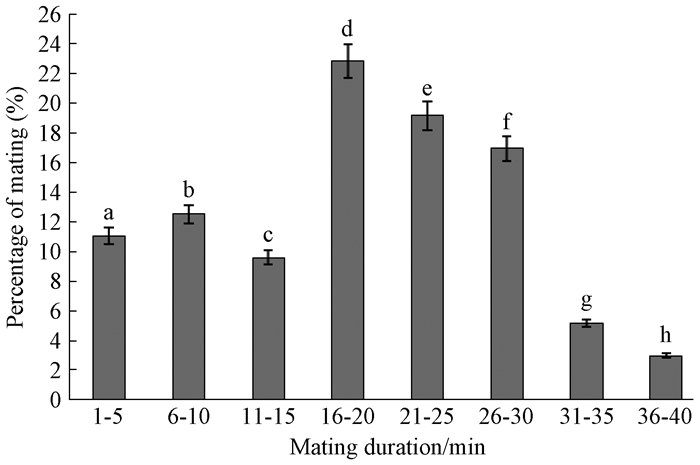
|
Fig.1 Mating duration of B.melanopus adults |
The highest mating frequency occurred at 10:00, with less peaks at 16:00 and 18:00.At other times mating frequency remained low.However, there were no significant differences among times in the circadian rhythm of mating(df=11, P=0.000)(Fig. 2).

|
Fig.2 Daily mating pattern of B.melanopus adults The percentage is the mean value of a cumulative amount of three days, from three repeats(mean ±SE); the bars followed by the different letters are statistically different between two different treatments at P≤0.05 level according to ANOVA:LED test; the mating rate was the average rate of mating times of total number of times within 2 h in 3 d. |
Ovipositing peaked from 16:00 to 18:00, with lower levels of activity at 14:00, 22:00 and at 2:00.The mean number of eggs of per female was (275.5±131.4) eggs(df=11, P=0.000)(Fig. 3).
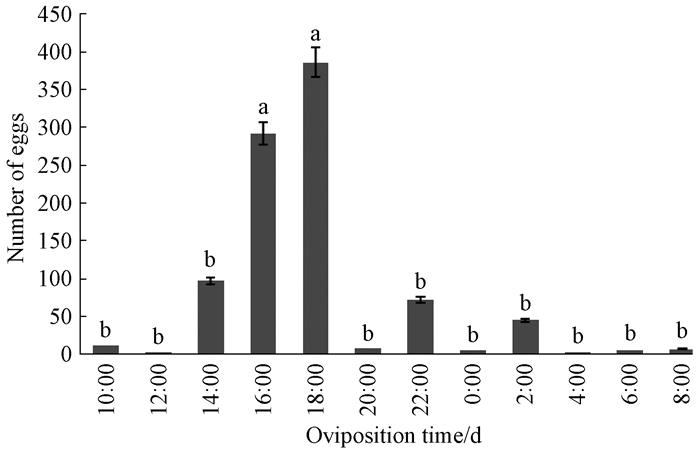
|
Fig.3 Daily oviposition rhythm of B.melanopus adults The number of eggs laid was the mean value of a cumulative amount of three days, from three repeats(mean ±SE); the bars followed by the different letters are statistically different between two different treatments at P≤0.05 level according to ANOVA:LED test. |
We found significant differences in the duration(P=0.010) of the egg stage between 23, 26, and 29 ℃.Generally, the duration of the egg stage shortened with increasing temperature.Meanwhile, hatching rates were not high at any temperature.The highest hatching rate was just 27.0% at 26 ℃, and less than 5%at 23 ℃ and 29 ℃.Overall larval mortality reached 100%, which suggests that temperature had no effect on hatching rate(P=0.160).A shortage of artificial diet may account for these results.More than 82% of eggs were laid during the first 20 days, and the number of ovipositions decreased over time.
Oviposition peaked from day 7-9 and then decreased quickly on day 10 on.Mean egg production per female was(275.5±131.4)eggs(Fig. 4).
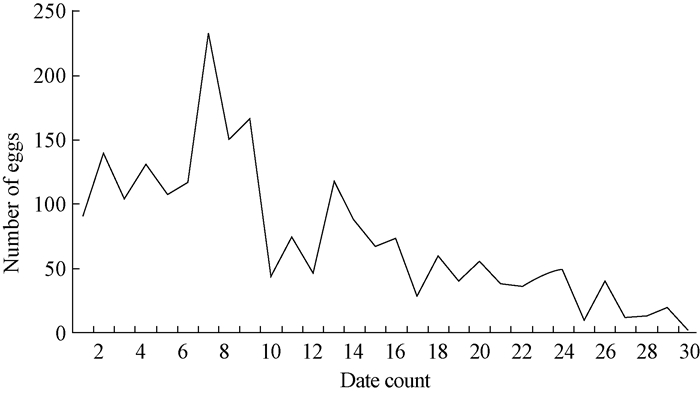
|
Fig.4 Oviposition rhythm of B.melanopus |
The smaller males are preferred(57.4%) by females, but larger males also had chances to mate(42.6%;P=0.026)(Fig. 5).

|
Fig.5 Mating choices of female to males with different body size under long-term paired condition The bars followed by the same letters are statistically indifferent between two different treatments at P≤0.05 level according to ANOVA:LED test.The large(6.0-6.5 mg) in black and the small one(4.1-4.5 mg) in grey. |
|
|
Males, meanwhile, chose most often(52.6%) to mate with large females, although this choice is not significantly different(P=0.574;Fig. 6).
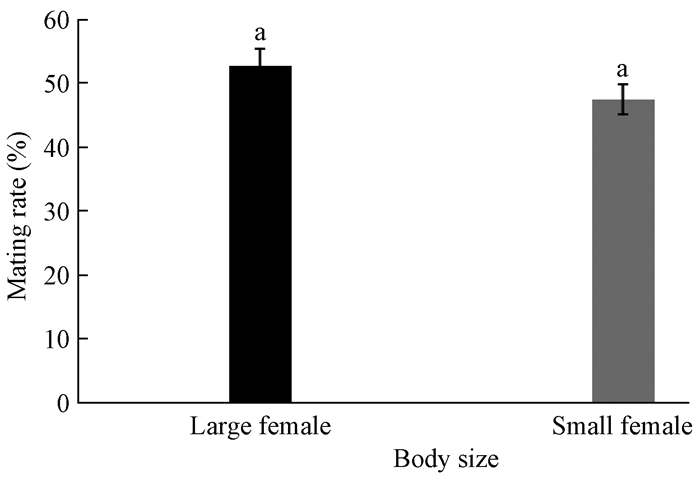
|
Fig.6 Mating behaviour of males with females with different body size under long-term paired condition The bars followed by the same letters are statistically indifferent between two different treatments at P≤0.05 level according to ANOVA:LED test.The large female(6.1-7.3 mg) in black and the small one(4.2-5.0 mg) in grey. |
Except for day two(P=0.191) virgin females preferred to mate with a virgin male compared to a mated male on the 1st and 5th day.However, the preference is reversed on the 3rd and 4th day.In the aggregate, the preferences of the virgin female to the mated and virgin males are roughly the same.So, this indicates that there is no significant difference of male mating experience on the sexual selection of female (df=8, P=0.307)(Fig. 7).
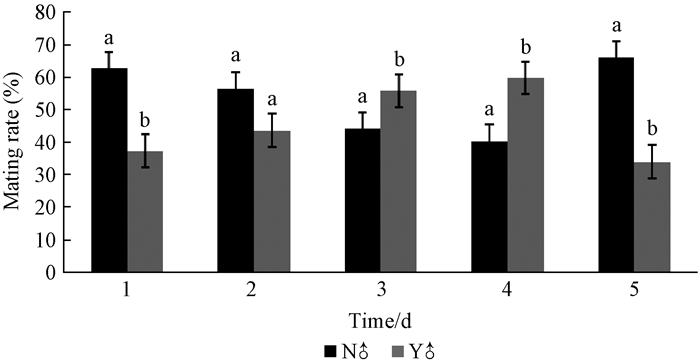
|
Fig.7 The effect of mating experience of males on females N♂ means virgin male, Y♂ means male which mated once. Different letter on the bar in one day indicate difference is significant with the selection of female between virgin male and mated male at 5% level. |
There is an extremely significant difference in the preference of virgin males' choice between virgin females and mated females(Fig. 8).And the preferences of the virgin male choosing mated and virgin females are roughly the same on the 1st day (P=0.803), while on the other 4 days, the virgin males significantly preferred mated females.It shows that the mating experience of female has a dominant influence on the selection of the male(df=8, P=0.001).
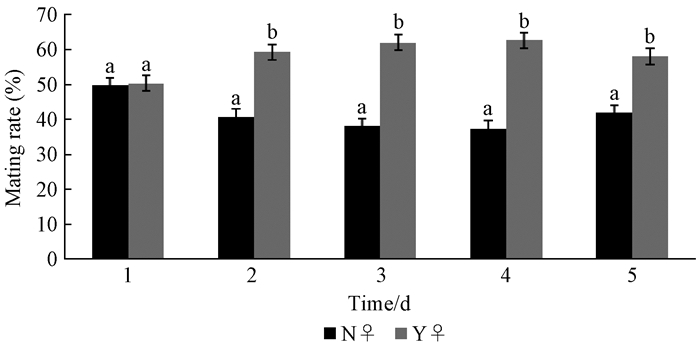
|
Fig.8 The effect of mating experience of females on males N♀ means virgin female, Y♀ means female which mated once. Different letter on the bar in one day indicate difference is significant with the selection of male between virgin female and mated female at 5% level. |
In this study, female B.melanopus beetles oviposited for up to 30 days, and the mean egg production per female was(275.5±131.4), findings different from those of Tan et al.(1986), who observed an oviposition period of only 15 days and egg-production per female of 50-80 eggs.These differences may be due to environmental conditions, different host plants(B.melanopus wasfed on C.oleifera in our study, but C.sinensis in the other study) or different test procedures.Overall mortality was 100%in our study, which may be due to environmental conditions or to a lack of suitable artificial diet for the larvae.
We found B.melanopus to mate for 16-30 min, with the maximum duration under one hour, but some insects can mate for hours while others copulate for only a few minutes(Zhang et al., 2006; Han et al., 2000).The longest mating of an insect such as Jadera haematoloma may reach 11 days (Carroll, 1991) and the mating duration of the same species may be different under different conditions, environments or at different ages(Guo, 2010; He et al., 2015; Jersabek et al., 2007; Zhang et al., 2012).B.melanopus can mate at any time of day, but showed a primary mating peak at 10:00 and a lower one at 16:00-18:00, while daily oviposition peaked at 16:00-18:00.In insects, the female typically chooses the male, and while the male sometimes chooses the female, this is rare(Yang et al., 2013).Similar to our study, female Octodonta nipae were found to choose smaller males for a mate under long-term paired conditions(Chen, 2012), but females from other species have been found to prefer to mate with bigger males, such as the Phoracantha semipunctata (Hanks et al., 1996) and Menochilus sexmaculatus(Dubey et al., 2016).These preferences may be related to external factors and other conditions of the males, however.For example, males with wider upper prothoraxes were more likely to be selected by female Galerucella nymphaeae(Parri et al., 1998), while female Zorion guttigerum will choose mates based on a combination of body size and color(Qiao, 2002).However, when body size is significantly influenced by temperature variation, it is not correlated with male mating success(Pavkovic-Lucic et al., 2013) among protein-fed males of Ceratitis capitata, large males were more likely to copulate than small males(Kaspi et al., 2000), suggesting that nutritional resources are one of the important factors in sexual selection.Mating experience is another influencing factor in most Chrysomelidae like Galerucella birmanica(Fan, 2015).While female B.melanopus showed no significant selection based on the male's mating experience, females of the Lobesia botrana are able to discriminate between males with different mating experience, and prefer virgin males(Muller et al., 2016), while as shown in Ophraella communa, male B.melanopus prefers to mate with a female that has had mating experience(Guo, 2010).
B.melanopus laid eggs mainly in the first 20 days of the oviposition period, with a distinct peak period.This is similar to the oviposition trend of Harmonia axyridis, suggesting the existence of an optimal number of matings(Xiao, 2010).While the hatch rate of eggs in our study was very low, this was probably because of a difference between the temperature in the laboratory and that of B.melanopus habitat in the field combined with an additional bacterial infection.
While it is clear that B.melanopus adultscan mate and oviposit many times during their lifetimes, the links between multiple matings and ovipositing, the influence of temperature, age, and lighting on mating and oviposition, and the effects of mating on the hatch rate of eggs all need to be further explored.Our study is but a first step toward improving our ability to forecast B.melanopus population growth and control its outbreaks.
Carroll S P. 1991. The adaptive significance of mate guarding in the soapberry bug, Jadera haematoloma(Hemiptera:Rhopalidae)[J]. Journal of Insect Behavior, 4(4): 509-530. DOI:10.1007/BF01049335 |
Chen M M. 2012. Multiple-mating behavior and its reproductive benefit on Octodontanipea[J]. Fuzhou:MS thesis of Fujian Agriculture and Forestry University. |
Dubey A, Omkar, Mishra G, et al. 2016. Adult body size drives sexual selection mutually in the ladybird, Menochilus sexmaculatus(Coleoptera:Coccinellidae)[J]. Acta Entomologica Sinica, 59(2): 209-218. |
Fan H J. 2015. Mating Behaviour of Galerucella birmanica Jacoby[J]. Wuhan:MS thesis of Huazhong Agricultural University. |
Guo W. 2010. Behavioral strategy of mating and oviposition of adult Ophraella communa[J]. MS thesis, Hunan Agricultural University. |
Han G B, Du J W, Li J. 2000. Mating behavioral ecology of Ancylis sativa adult[J]. Chinese Journal of Applied Ecology, 11(1): 99-102. |
Hanks L M, Millar J G, Paine T D. 1996. Body size influences mating success of the Eucalyptus long-horned borer(Coleoptera:Cerambycidae)[J]. Journal of Insect Behavior, 9(3): 369-382. DOI:10.1007/BF02214016 |
He H M, Xiao L, Fu S, et al. 2015. Observations on the life history, mating and fecundity of Platycorynus parryi Baly[J]. Chinese Journal of Applied Entomology, 52(1): 193-199. |
He X Y, Cai S P, Wu Z C. 2011. Basilepta melanopus Lefèvre, a new insect pest in Camellia oleifera[J]. Forest Pest and Disease, 30(3): 16-17. |
Jersabek C D, Luger M S, Schabetsberger R. 2007. Hang on or run? Copepod mating versus predation risk in contrasting environments[J]. Oecologia, 153(3): 761-773. DOI:10.1007/s00442-007-0768-1 |
Jie Y Z, Zhao D Y, Qin C S, et al. 2014. Present situation and controlling measures on Camellia gauchowensis pest and disease[J]. Guangdong Forestry Science and Technology, 30(5): 24-28. |
Kaspi R, Taylor P W, Yuval B. 2000. Diet and size influence sexual advertisement and copulatory success of males in Mediterranean fruit fly leks[J]. Ecological Entomology, 25(3): 279-284. DOI:10.1046/j.1365-2311.2000.00266.x |
Li M, He Z, Xia Y G, et al. 2013. Occurrence and control of Basilepta melanopus Lefèvre in main oil Camellia production area of Hunan Province[J]. Forest Pest and Disease, 32(2): 32-35. |
Li M, Zhou G, He Z, et al. 2014. Spatial distribution patterns and the environmental interpretations of Basilepta melanopus(Coleoptera:Eumolpidae) adults in Camellia oleifera young forests[J]. Scientia Silvae Sinicae, 50(10): 173-180. |
Muller K, Arenas L, Thiéry D, et al. 2016. Direct benefits from choosing a virgin male in the European grapevine moth, Lobesia botrana[J]. Animal Behaviour, 114: 165-172. DOI:10.1016/j.anbehav.2016.02.005 |
Parri S, Alatalo R V, Mappes J. 1998. Do female leaf beetles Galerucella nymphaeae choose their mates and does it matter?[J]. Oecologia, 114(1): 127-132. DOI:10.1007/s004420050428 |
Pavkovic-Lucic S, Kekic V. 2013. Developmental temperature, body size and male mating success in fruit flies, Drosophila melanogaster(Diptera:Drosophilidae)[J]. European Journal of Entomology, 110(1): 31-37. DOI:10.14411/eje.2013.004 |
Qiao W. 2002. Sexual selection of Zorion guttigerum Westwood(Coleoptera:Cerambycidae:Cerambycinae) in relation to body size and color[J]. Journal of Insect Behavior, 15(5): 675-687. DOI:10.1023/A:1020747907490 |
Tan J C, Liu G F, Wang D X, et al. 1986. Study on the biological characteristics and control of Basilepta melanopus Lefèvre[J]. Journal of Hunan Agricultural College, (4): 51-60. |
Wang R Z, Hu Y. 2004. The Occurrence and Control of Basilepta melanopus Lefèvre in Jiangxi[J]. Jiangxi Plant Protection, 27(4): 175. |
Xiao H. 2010. Study on multiple-mating and fitness of Harmonia axyridis[J]. MS thesis of Shanxi Normal University. |
Xu S B. 2001. Regression analysis of production function model and its application[J]. MS thesis of Xiangtan University. |
Yang X L, He Z R. 2013. Sexual selection and speciation[J]. Journal of Yunnan University, 35(S2): 364-367. |
Zeng M S, Liu F J, Wang D F, et al. 2012. An experimental demonstration of comprehensive control for Basilepta melanopus Lefèvre[J]. Fujian Journal of Agricultural Sciences, 27(8): 847-852. |
Zhang Q Q, Zhang X L, Lu W. 2012. Effects of adult age and mating status on fecundity of Haritalodes derogata[J]. Plant Protection, 38(2): 71-74. |
Zhang Y H, Hao D J, Wang Z, et al. 2006. The mating and ovipositing behavior of Monochamus alternatus[J]. Chinese Bulletin of Entomology, 43(1): 47-49. |
Zhu L J. 2013. The occurrence causes and countermeasures of Basilepta melanopus Lefèvre in Zhaoping County[J]. Agricultural Research and Application, (2): 64-67. |
 2018, Vol. 54
2018, Vol. 54

