文章信息
- Xu Xiaoniu, Wang Qin, Eiji Hirata
- 徐小牛, 王勤, 平田永二
- Effects of Broad-leaved Trees Invasion on Organic Carbon and Nitrogen Dynamics in Pinus Luchuensis Plantation
- 自然侵入阔叶树对琉球松人工林碳素及氮素动态的影响
- Scientia Silvae Sinicae, 2004, 40(1): 11-17.
- 林业科学, 2004, 40(1): 11-17.
-
文章历史
Received date: 2002-05-08
-
作者相关文章
2. 日本琉球大学农学部 冲绳 903-0213
2. Faculty of Agriculture, University of the Ryukyus, Nishihara Okinawa 903-0213, Japan
Soil organic matter in surface mineral soil is believed to be a major determinant of site productivity within many forest ecosystems due to its effects on soil structure, nutrients supplies (Powers et al., 1990). Likewise, the availability of soil N is the dominant limited nutrient in many forests, which has a major influence on primary productivity, species composition and species diversity in terrestrial ecosystems (Pastor et al., 1984; Wedin et al., 1990). The supply rate of N depends largely on N mineralization, which is usually regulated by both abiotic and biotic factors (Dwyer et al., 1981; Bosatta et al., 1982; Birk et al., 1986; Cornejo et al., 1994). Of the biotic factors, the supply of litterfall is the most important, because litterfall can differ widely in decomposition rate depending on the quality, quantity, and timing of inputs (McClaugherty et al., 1987; Berendse et al., 1989; Murdick et al., 1994). Thus, the nature of the vegetation can have a large effect on the N supply rate in an ecosystem.
The objectives of this study are to determine the amount and variation of organic C and total N in the forest floor and soil for different stand types and to examine the effect of broad-leaved trees invasion on soil and litterfall nitrogen dynamics in Pinus luchuensis plantation.
1 Methods 1.1 Study siteThe study site is located in Yona Experimental Forest of the University of the Ryukyus, northern part of Okinawa Island, Southwest Japan. The latitude and longitude of the site are 26°45′30″N and 128°05′E, respectively. The climate is warm and humid, with a mean annual air temperature of 21.6 ℃ and a mean annual precipitation of 2 660 mm. The mean minimum air temperature is 10.8 ℃ in January (Yona Experimental Forest).
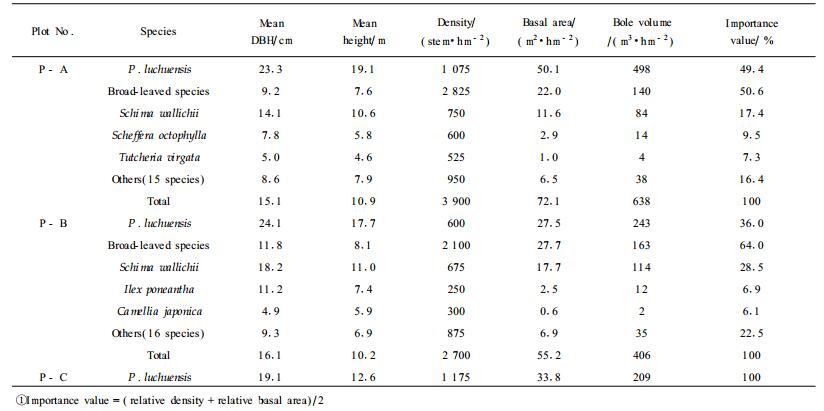
A total of 16 hm2 P. luchuensis plantations was established there in the 1950s. Most of those plantations were invaded by volunteer broad-leaved trees, especially by Schima wallichii after repeatedly severe disturbance of typhoons which caused about 10%~20% of the individuals to breakdown and to uproot in the 1970s. The forest structure differed with the disturbance regime and topographical feature. Additionally, those stands had not received any management treatments such as logging or thinning since 1980s. Three 20 m×20 m sampling plots were set up, of which one was in the pure pine stand (P-C) and two in the pine-broadleaved tree mixture (P-A and P-B). The general description of the structures for the sampling stands and their environment is given in Tab. 1. A multi-story structure was formed in the mixed stand, within which pine trees occupied the upper canopy (15~20 m high), and sub-dominant, S. wallichii occupied the sub-canopy layer (8~15 m high). The soil on the site is yellow soil (Forest Soil Division, GESF, 1976) developed from tertiary sandstone (Yamamori, 1994).
|
|
After plot installation, the forest floor and mineral soil were, respectively, sampled in February, June and October 1997, from six 1 m×1 m quadrats randomly located within each plot. L, and F + H layers were lifted from each quadrat. The composition of L layer was sorted into leaf, small wood (including branches and barks less than 2.0 cm in diameter), and sexual organs (including flower, fruit and cone). Their wet weights were measured in the field. Litter from the six quadrats was thoroughly mixed to give one litter composite per plot; and partial samples were extracted for dry weight determination and chemical analysis. Mineral soil to 30 cm was extracted in each quadrat with a cylindrical sampler with an inner diameter of 10 cm. Soil samples were oven-dried at 70℃ to a constant weight and ground to pass through a 2 mm sieve prior to analysis.
1.3 Litterfall collectionLitterfall was collected in the pure pine and pine-broadleaved tree plots. Five litter traps with an opening of 1.2 m in diameter were placed in each plot. Litterfall was collected monthly from August 1996 to July 1999. Litter was sorted by tissue type (needle, leaf, small wood, sexual organ, and miscellaneous), dried at 70℃ to a constant mass, then weighed. Subsamples from each plot were ground for chemical analysis.
1.4 Mineralization experimentTo determine if differences in soil N mineralization among plots were related to species composition or litter quality, we conducted a 30-day laboratory incubation study, which involved three plots, one soil layer, and five laboratory replications. In July 1997, mineral soil was resampled at six random points for each plot. Mineral soil was extracted at each point with a cylindrical sampler for 0~10 cm layer, and delivered to the laboratory soon. The fresh soil samples from six sampling points were passed through a 2 mm sieve, and thoroughly mixed. Ten 20 g subsamples were taken from each of the composite samples. Two subsample were for moisture determination, three for initial mineralizable N determination, and five for 30 d of incubation at 25℃. The soil samples for incubation were placed in a 100 mL polyethylene bottle. The mouth of the bottle was sealed with gas-permeable plastic. Before incubation, the soil moisture of each subsample was adjusted to field capacity using deionized, distilled water. We chose a soil moisture of field capacity and a soil temperature of 25℃ to simulate optimal soil environmental conditions.
1.5 Chemical analysis and data treatmentSoil pH was determined in a 1:2 soil/water mixture by glass electrode. Total organic carbon and total nitrogen in forest floor and soil samples were determined by dry combustion with a C-N Corder (Yanaco, MT-500, Kyoto, Japan). The mineralizable N (NH4++NO3-) in initial and incubated subsamples were extracted by adding 100 mL of 2mol·L-1 KCl, shaking for 2 h and extracting for an additional 22 h. The extract was filtered and analyzed for NH4+ and NO3- concentrations by steam distillation (Hidaka, 1997). Net mineralization was calculated as the difference between (NH4++NO3-) concentrations of the incubated and initial soil samples.
In order to ascertain whether C and N dynamics differed significantly between the pure and mixed pine stands, t-test was used, and significance was accepted at p=0.05.
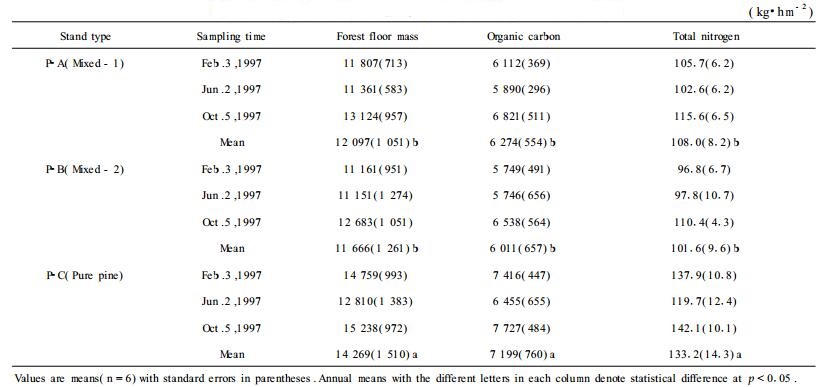
2 Results 2.1 Forest floor organic carbon and nitrogenForest floor accumulation averaged 14.2 Mg·hm-2 in the pure pine stand (P-C) with an average of 7.2 Mg C·hm-2 and 133 kg N·hm-2 (Tab. 2). Forest floor in the mixed stands (P-A and P-B) was about 11.9 Mg·hm-2. The accumulations of organic C and total N in the forest floor were significantly lower under the mixed stands than the pure pine stand (p < 0.001). Differences within stand for different sampling times were also found for organic C and N in forest floor (p < 0.05). Generally, higher accumulation occurred in October. It was due to the effect of typhoons which caused 3.0~4.8 Mg·hm-2 of litterfall in August 1997 (Xu et al., 2002).
|
|
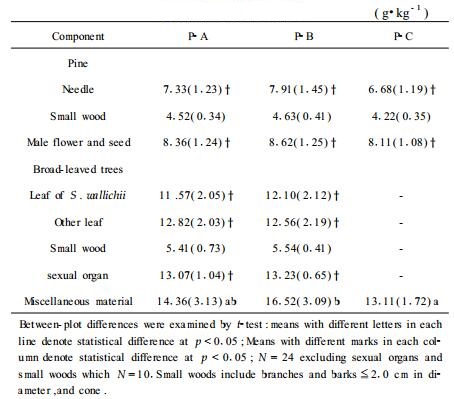
Needle litterfall N concentration for P. luchuensis pine ranged from 6.7 g·kg-1 in pure stand (P-C) to 7.9 g·kg-1 in the mixed one (P-B), and for the subdominant, S. wallichii, ranged from 11.6 g·kg-1 in the P-A to 12.1 g·kg-1 in the P-B (Tab. 3). N concentration of leaf litterfall differed significantly (p < 0.001) between the two main species, P. luchuensis and S. wallichii; however, no differences were found for the same species between stand types and between sampling years. Again, N concentration of miscellaneous material was significantly greater (p < 0.05) in the P-B than in P-A and P-C (Tab. 3), which could be attributed to the different composition between the pure and mixed pine stands.
|
|
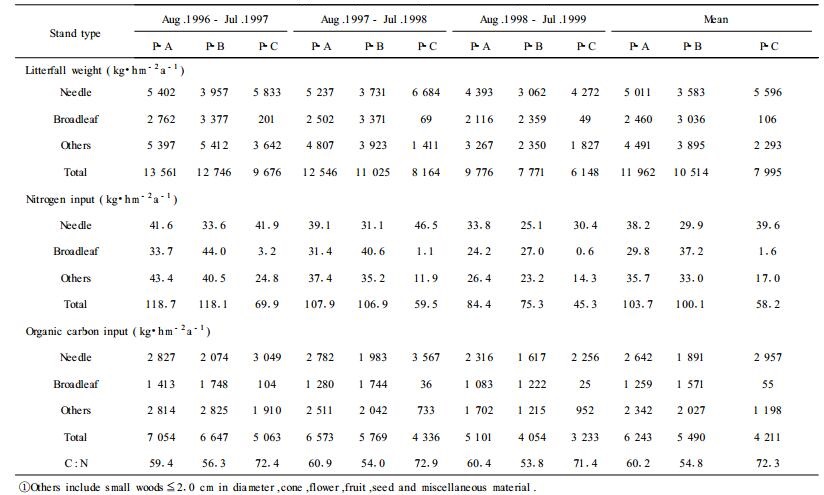
The annual N input by litterfall averaged 58.2 kg N·hm-2 in the pure pine stand (P-C), which was significantly lower (p < 0.01) than that in the mixed stands (104 kg N·hm-2 for P-A and 100 kg N·hm-2 for P-B; Tab. 4). Within either the pure or the mixed stand, N content was significantly lower in the third year than the first two years, but not significantly different between the first and second years (p > 0.143).
|
|
The annual C input by litterfall averaged 6 243 kg·hm-2, 5 490 kg·hm-2 and 4 211 kg·hm-2 for P-A, P-B, and P-C, respectively (Tab. 4). And the mixed stands were significantly greater than the pure pine stand (p < 0.05).
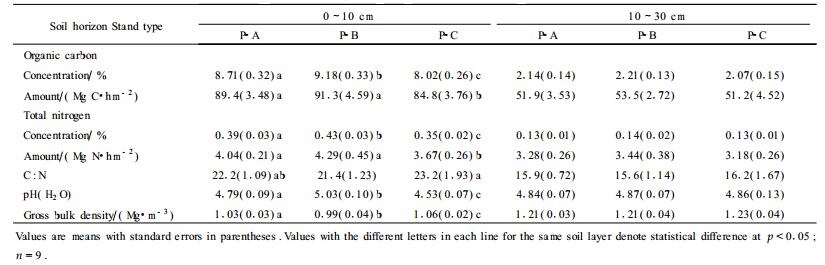
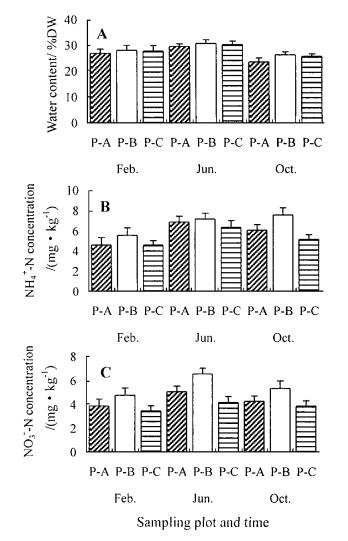
2.3 Soil nitrogen dynamicsSeasonal patterns for soil moisture and NO3- and NH4+ concentrations are shown in Fig. 1. Except for P-A in October, soil moisture generally did not differ significantly (p > 0.05) among stand types. Soil NO3- and NH4+ concentrations differed significantly (p < 0.05) among stand types. Mean NH4+ and NO3- concentrations (mg·kg-1) in the top 10 cm mineral soil were, respectively, 5.4 mg·kg-1 and 3.9 mg·kg-1 for the pure pine stand (P-C), 5.9 mg·kg-1 and 4.4 mg·kg-1 for P-A, and 6.8 mg·kg-1 and 5.6 mg·kg-1 for P-B.
|
Fig.1 Seasonal percentage of soil moisture(A), and NH4+(B)and NO3-(C) concentrations in the top 10 cm of mineral soil for the sampling plots under Pinus liukiuensis plantations.Vertical bar is one standard error. |
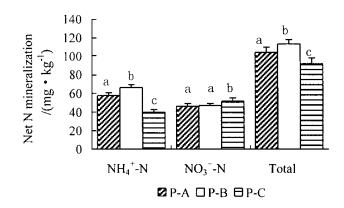
N mineralization rate of the top 10 cm mineral soil during 30-day incubation under the mixed stands was significantly higher than under the pure pine stand(p < 0.01). Significant differences were apparent among the sampling plots (Fig. 2).
|
Fig.2 Net N mineralization of the top 10 cm mineral soil in 30-day incubation for the sampling plots in luchu pine plantation.Net N mineralization rates with same letters do not differ significantly (p>0.05) among the sampling plots.Vertical bar is one standard error. |
Total N concentrations for the fine soil (≤2 mm particles) in 0~10 cm under the mixed stands (P-A and P-B) were significantly higher (p < 0.01) than that under the pure pine stand (P-C; Tab. 5). For 10~30 cm soil layer, Total N concentrations showed little or no stand type difference. Like soil N, soil organic C in the mixed stands was higher than that in the pure pine stand, but the differences were only in the top 10 cm soil layer (Tab. 5).
|
|
In this study, the forest floor N content was greater in the pure pine stand than in the mixed stands (Tab. 2). However, in the top 10 cm of mineral soil, the mixed stands had an average of 493 kg N·hm-2 more than the pure stand did (Tab. 5). Because of no pretreatment measurements, we cannot rule out the possibility of inherent site differences. However, the relative uniformity of environment and soil type within the study area suggests that differences are probably related to the volunteer broad-leaved trees, which can either accelerate N turnover in the forest floor (Takahashi, 2000) or cause difference in N distribution rather than in total N within the system (Alban, 1982). The present study demonstrated that the significant effect of broad-leaved trees on N accumulation was limited to the top 10 cm mineral soil layer (Tab. 5). This is in agreement upon the result from Lefevre and Klemmedson (1980).
Again, in pine-broadleaved tree mixture, the net rate of soil N mineralization averaged 18 % greater than that in the pure pine stand (Fig. 2). A higher mineral N concentration of surface mineral soil was also present in pine-broadleaf mixture (Fig. 1B and 1C). These results are interpreted as the influence of volunteer broad-leaved trees dominated by S. wallichii, on the accumulation and cycling of N. In the mixed stands, the concentration of total N in broadleaf litter was about 62% greater than in the pine needle litter (Tab. 3), and broadleaf litter reached an average of 39 % of the annual total leaf litter. Thus, the annual N input in litterfall in the mixed stands was 1.7 times that in the pure pine stand. The greater annual litterfall N input and relatively lower C:N ratio in the mixed stands may be correlated to the higher net N mineralization in forest floor and mineral soil (Pastor et al., 1984; McNulty et al., 1991; Reich et al., 1997; Prescott et al., 2000). Some other studies have demonstrated a vegetation effect on soil N mineralization (Wedin et al., 1990; Gower et al., 1992; Compton et al., 1998; Hirai, 1999). We do not have adequate pretreatment data to conclusively demonstrate that soil N was similar within each sampling stand, but several indirect lines of evidence suggest differences were not apparent. The results from this study showed the importance of stand type (or species composition) differences in determination of organic matter quality in affecting N cycling, particularly the difference between conifer and broadleaf, and between-species interaction (Klemmedson, 1987; Perry et al., 1987). Our previous research has showed that leaf litter of S. wallichii decomposed much faster than the pine needle (Xu et al., 2000). After two years' decomposition, leaves of S. wallichii disappeared completely, while the pine needles lost only 40% of total N and 55% of organic C. Thus, net N release may be strongly affected by species for they affect on litter quality. More rapid turnover of total N and organic C in forest floor and mineral soil is expected in the mixed stands because of the presence of the relatively favorable quality of broadleaf litter.
Annual N input in litterfall in this study ranged from 58.2 kg N·hm-2 in the pure pine stand to 103.7 kg N·hm-2 in the mixed stand (P-A) over 3 years, which were much higher than warm temperate coniferous forests (Vogt et al., 1986; Tsutsumi, 1987). High annual N input in litterfall for the mixed stand may be contributed to the different structural characteristics from the pure pine stand. A multi-story structure was formed in the pine-broadleaved mixture, which is good to generate beneficial microclimate, and to increase stand productivity (Xu etal., 1997). Thus, the mixed stand appears high productivity (Xu et al., 2002), which contributes to the greater annual rates of litterfall and related N input.
Acknowledgement
We thank Dr. N. Yamamori, emeritus professor of the University of the Ryukyus for his advice and invaluable comments. We also thank the staff of Yona Experimental Forest, University of the Ryukyus, for their support to field and laboratory works. This study was made possible by partial support from the Japanese Ministry of Education, Science, Sports, and Culture grant to X.N. Xu.
Alban D H. 1982. Effects of nutrient accumulation by aspen, spruce, and pine on soil properties. Soil Sci Soc Am J, 46: 853-861. DOI:10.2136/sssaj1982.03615995004600040037x |
Berendse F, Bobbink R, Rouwenhorst G. 1989. A comparative study on nutrient cycling in wet heathland ecosystems. Ⅱ. Litter decomposition and nutrient mineralization. Oecologia, 78: 338-348. DOI:10.1007/BF00379107 |
Birk E M, Vitousek P M. 1986. Nitrogen availability and nitrogen use efficiency in loblolly pine stands. Ecology, 67: 69-79. DOI:10.2307/1938504 |
Bosatta E, Staaf H. 1982. The control of nitrogen turnover in forest floor. Oikos, 39: 143-151. DOI:10.2307/3544478 |
Compton J E, Boone R D, Motzkin G., et al. 1998. Soil carbon and nitrogen in a pine-oak sand plain in central Massachusetts: Role of vegetation and land-use history. Oecologia, 116: 536-542. DOI:10.1007/s004420050619 |
Cornejo F H, Varela A, Wright S J. 1994. Tropical forest litter decomposition under seasonal drought: nutrient release, fungi, and bacteria. Oikos, 70: 83-90. |
Dwyer L M, Merriam G. 1981. Influence of topographic heterogeneity on deciduous litter decomposition. Oikos, 37: 228-237. DOI:10.2307/3544470 |
Forest Soil Division, GF ES. 1976. Forest soil classification in Japan. Bull Govt For Exp Sta, 280: 1-28. |
Gower S T, Son Y. 1992. Differences in soil and leaf litterfall nitrogen dynamics for five forest plantations. Soil Sci Soc Am J, 56: 1959-1966. DOI:10.2136/sssaj1992.03615995005600060051x |
Hidaka S. Nitrogen. In Committee of Methods for Soil Environment Analysis (ed.), Methods for soil environment analysis. Tokyo: Hakuyusha, 1997: 225-231(in Japanese)
|
Hirai K. 1999. Nitrogen mineralization of undisturbed samples in some forest soils. Jpn J Soil Sci Plant Nutr, 70: 324-327. |
Klemmedson J O. 1987. Influence of oak in pine forests of central Arizona on selected nutrients of forest floor and soil. Soil Sci Soc Am J, 51: 1623-1628. DOI:10.2136/sssaj1987.03615995005100060039x |
Lefever R E, Klemmedson J O. 1980. Effect of Gambel oak on forest floor and soil of a ponderosa pine forest. Soil Sci Soc Am J, 44: 842-846. DOI:10.2136/sssaj1980.03615995004400040037x |
McClaugherty C, Berg B. 1987. Cellulose, lignin and nitrogen concentrations as rate regulating factors in late stages of forest litter decomposition. Pedobiologia, 30: 101-112. |
McNulty S G, Aber D J, Boone R D. 1991. Spatial changes in forest floor and foliar chemistry of spruce-fir forests across New England. Biogeochemistry, 14: 13-29. |
Murdick D A, Hoosein M, Hicks R P., et al. 1994. Decomposition of leaf litter in an Appalachian forest: effects of leaf species, aspects, slope position and time. For Ecol Manage, 68: 231-250. DOI:10.1016/0378-1127(94)90048-5 |
Paster J, Aber J D, McClaugherty C A., et al. 1984. Aboveground production and N and P cycling along a mineralization gradient on Blackhawk Island, Wisconsin. Ecology, 65: 256-268. DOI:10.2307/1939478 |
Perry D A, Choquette C, Schroeder P. 1987. Nitrogen dynamics in conifer-dominated forests with and without hardwoods. Can J For Res, 17: 1434-1441. DOI:10.1139/x87-221 |
Power R F, Allan D H, Miller R E. et al. Sustaining site productivity in North American forests: Problems and prospects. In Gessel S P. et al. (eds.) Sustained productivity of forest soils. Proc North Am For Soils Conf 7th, Univ of Br Columbia, Vancouver. 1990: 49-79
|
Prescott C E, Chappell H N, Vesterdal L. 2000. Nitrogen turnover in forest floors of coastal Douglas-fir at sites differing in soil nitrogen capital. Ecology, 81: 1878-1886. DOI:10.1890/0012-9658(2000)081[1878:NTIFFO]2.0.CO;2 |
Reich P B, Grigal D F, Aber J D., et al. 1997. Nitrogen mineralization and productivity in 50 hardwood and conifer stands on diverse soils. Ecology, 78: 335-347. DOI:10.1890/0012-9658(1997)078[0335:NMAPIH]2.0.CO;2 |
Takahashi T. 2000. Effects of supply of deciduous broad-leaved litter to the forest floor on the soil fertility of a Japanese cypress (Chamaecyparis obtuse) forest. Jpn J For Environ, 42: 23-28. |
Tsutsumi T. 1987. Nutrient cycling in forest ecosystems. Tokyo: Tokyo Univ Press: 124.
|
Vogt K A, Grier C C, Vogt D J. 1986. Production, turnover, and nutrient dynamics of above-and below-ground detritus of world forests. Adv Ecol Res, 15: 303-366. DOI:10.1016/S0065-2504(08)60122-1 |
Wedin D A, Tilman D. 1990. Species effects on nitrogen cycling: a test with perennial grasses. Oecologia, 84: 433-441. DOI:10.1007/BF00328157 |
Xu X N, Li H K. 1997. Study on the growth of the mixed stand of Pinus massoniana and Liquidambar formosana and its ecological benefits. Scientia Silvae Sinicae, 33: 385-393. |
Xu X N, Hirata E. 2002. Forest floor mass and litterfall in luchu pine plantations with and without broad-leaved trees. For Ecol Manage, 157: 165-173. DOI:10.1016/S0378-1127(00)00663-0 |
Xu X N, Hirata E, Enoki T., et al. 2000. Leaf litter decomposition of three selected species in subtropical forests at northern Okinawa, Japan. Trans Ann Mtg Jpn For Soc, 111: 102-103. |
Yamamori N. 1994. Environmental conditions of Yona Experimental Forest. In the 40th anniversary of the foundation of university forest, Fac Agric Univ Ryukyus: 35-53. |
 2004, Vol. 40
2004, Vol. 40