2. Department of Urology, General Hospital of Ningxia Medical University, Yinchuan 750004, Ningxia, China;
3. Department of Obstetrics and Gynaecology, Peking University Third Hospital, Beijing 100191, China;
We used Smo siRNA to inhibit hedgehog signaling pathway in embryonic day (E) 13 palatal shelves in organ culture. SiRNA 4 was chosen as the most efficient from four synthesized Smo siRNAs. Palatal shelf fusion rate of 4 μg/mL cyclopamine group was the lowest and significantly lower than that of blank control group (P<0.05), and that of siRNA 4 group was also lower than that of blank control group (P=0.183). At 48 h after transfection, Smo protein level of siRNA 4 group was 64.8% lower than that of blank control group (P<0.05), and Gli1 protein level of 4 μg/mL cyclopamine group was 68.9% lower than that of blank control group (P<0.05). Hedgehog signaling pathway inhibition decreased palatal fusion in organ culture, probably owing to downregulation of Smo and Gli1 proteins.
Hedgehog signaling pathway plays a vital role in the process of embryonic development, including regulation of embryogenesis, tissue differentiation, and structure formation of brain, face, and other tissues. In vertebrates, hedgehog signaling pathway primarily comprises Hh ligands [Sonic Hh (Shh), Indian Hh (Ihh), and Desert Hh (Dhh)], a receptor complex composed of 12-transmembrane protein Patched (Ptc1 and Ptc2) and 7-transmembrane protein Smoothened (Smo), and nuclear transcriptional factor Gli family (Gli1, Gli2, and Gli3)[1]. The classic hedgehog pathway transduction starts with the binding of Hh ligands with Ptc1, which then unleashes Smo and further changes the structure of Gli transcription factors, finally resulting in the transcription of target genes[1]. However, in the absence of Hh ligands, Ptc1 inhibits the function of Smo[1].
In mice, the process of palatal genesis involves initiation (E11.5), vertical outgrowth (E13.5), elevation to horizontal position (E14.5), adhesion (~E15.0), and fusion (E15.5) of the bilateral palate[2]. Failure in any part of this process could cause a cleft palate[2]. The role of hedgehog signaling pathway in inducing cell proliferation and outgrowth of palatal shelves has been well established. Disruption of Fgf-Shh signaling network decreased cell proliferation in the palatal epithelium and mesenchyme, eventually causing cleft palate in mice[3]. Cre/loxP-mediated tissue inactivation of Smo gene in the developing mice palatal mesenchyme also reduced cell proliferation in the palatal epithelium and mesenchyme[4]. However, whether hedgehog signaling is involved in adhesion and/or fusion of palatal shelves is not well understood yet. Our hypothesis was that inhibition of hedgehog signaling pathway could impact palatal fusion. RNA interference (RNAi) is a newly developed technique that can decrease relevant gene expression by sequence-specific mRNA degradation guided by double-stranded RNA. In the present study, we utilized siRNA technique to knock down Smo gene expression in mice palatal culture at E13 to explore the influence of hedgehog signaling pathway inhibition on palatal fusion.
All animal procedures were approved by the Animal Care and Use Committee of Peking University. Palatal shelves were cultured according to the methods previously described[5]. Timed-pregnant ICR mice were sacrificed at E13. Three or four pairs of palatal shelves were placed into a 50-mL bottle containing 9 mL BGJb medium (GIBCO, Grand Island, NY, USA). Bottles containing the palatal shelves were sealed airtight after being flushed for 2.5 min with a gas mixture of 50% O2, 5% CO2, and 45% N2 using a needle and incubated at 37 °C in a roller device for 72 h at a speed of approximately 25 rpm. During the culture period, bottles were flushed every 24 h with the same gas mixture without medium exchange.
Three pairs of Smo siRNA and one pair of negative control siRNA were designed by Shanghai GenePharma Co., Ltd (Shanghai, China) in this study, namely siRNA 1059, siRNA 1306, siRNA 2073, and NC siRNA, respectively. Another pair of Smo siRNA drawn from a related research[6], namely siRNA 4, was added in the study. Table S1 (see the www.besjournal.com) displays the siRNA sequences. Then, 500 nmol/L siRNA aliquots were prepared by dissolving freeze-dried powder of siRNA in BGJb medium containing 0.2% OligofectamineTM reagent (Invitrogen, Carlsbad, CA, USA); the total volume was made up to 9 mL. The transfection was completed according to the standard procedure of OligofectamineTM reagent specification. Cyclopamine, a classic hedgehog pathway inhibitor, which could inhibit Smo from activation through binding with it directly[7], was used as the positive control and was dissolved in DMSO (Sigma, St. Louis, MO, USA).
Smo mRNA expression was determined by quantitative real-time PCR at 24 h after transfection. Total RNA was isolated from palatal shelves by Trizol (Invitrogen) and reverse-transcribed to cDNA using a reverse transcription system (Promega, Madison, WI, USA). Gene expression was quantified using the Taqman primers (Shanghai GenePharma Co., Ltd) and a real-time PCR machine (ABI PRISM®7500) from Applied Biosystems (Foster City, CA, USA). Quantitative protein analysis was performed using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE). Palatal shelves were harvested, respectively, at 24 h and 48 h after transfection. Total protein was isolated according to a modified radioimmunoprecipitation assay (RIPA) buffer protocol. Primary rabbit polyclonal antibodies against Smo (1:100), Gli1 (1:100), or β-actin (1:200) were purchased from Santa Cruz Biotechnologies (Santa Cruz, CA, USA). Primary rabbit polyclonal antibodies against Gli2 (1:200) and Gli3 (1:200) were purchased from Proteintech Group (Wuhan, China). Fusion of palatal shelves was observed under a stereomicroscope at 72 h after transfection. Fusion rate was calculated by dividing the total number of palatal shelves by the number of partially or completely fused palatal shelves. Data were analyzed using SPSS 13.0 statistical software. Results from multiple groups were compared using ANOVA and LSD/Tamhane’s T2 for multiple comparisons. Fusion rate was analyzed using χ2 test. Values of P<0.05 were considered statistically significant.
At 24 h after transfection, Smo mRNA expression showed no significant differences between siRNA-transfected groups (siRNA 1059 group, siRNA 1306 group, siRNA 2073 group) and blank control group (P>0.05), while that of siRNA 1059 group was the lowest (Figure S1, see the www.besjournal.com). Therefore, Western blot analysis was carried out to further confirm the influence of siRNA 1059 on Smo expression. However, the differences in Smo protein expression among siRNA 1059 group, NC siRNA group, and blank control group were not significant (P>0.05) at 24 h after transfection (Figure S2, see the www.besjournal.com). Comparisons of Smo protein levels at 24 h and 48 h after transfection among siRNA 1306 group, siRNA 2073 group, siRNA 4 group, NC siRNA group, and blank control group were further done by Western blot analysis (Figure S2, see the www.besjournal.com). Significant differences were found between each Smo siRNA group and blank control group at 48 h after transfection (P<0.05), but not at 24 h after transfection (P>0.05). Smo protein levels of siRNA 4 group, siRNA 1306 group, and siRNA 2073 group at 48 h after transfection were, respectively, 74.2%, 60.6%, and 36.1% lower than that of blank control group. Therefore, siRNA 4 was chosen as the most effective Smo siRNA and was used in the subsequent study. The obvious inhibition of Smo expression at 48 h rather than at 24 h by siRNA 4 after transfection might be related to factors such as Smo protein level and Smo half-life.
Palatal fusion was observed at 72 h after transfection; the results are displayed in Table 1. Palatal fusion rate of 4 μg/mL cyclopamine group was the lowest and was significantly lower than that of blank control group (P<0.05), while there were no differences in fusion rate between the remaining groups and blank control group (P>0.05). Palatal fusion rate of siRNA 4 group was lower than that of blank control group (χ2=1.769, P=0.183). These results indicated that siRNA 4 exhibited a trend of inhibiting palatal fusion, while 4 μg/mL cyclopamine completely affected this process. Altogether, these data suggest that hedgehog signaling pathway inhibition could disrupt palatal fusion.
|
|
Table 1 Effect of Smo siRNA and Cyclopamine on Palatal Fusion |
To investigate the possible mechanism involved, levels of hedgehog pathway proteins, Smo, and Gli1/2/3 were detected (Figures 1, 2). At 24 h after transfection, no significant differences in Smo level were found among the groups (P>0.05). At 48 h after transfection, Smo levels of siRNA 4 group, 4 μg/mL cyclopamine group, and 2 μg/mL cyclopamine group were, respectively, 64.8% (P<0.05), 49.5% (P>0.05), and 33% (P>0.05) lower than that of blank control group.
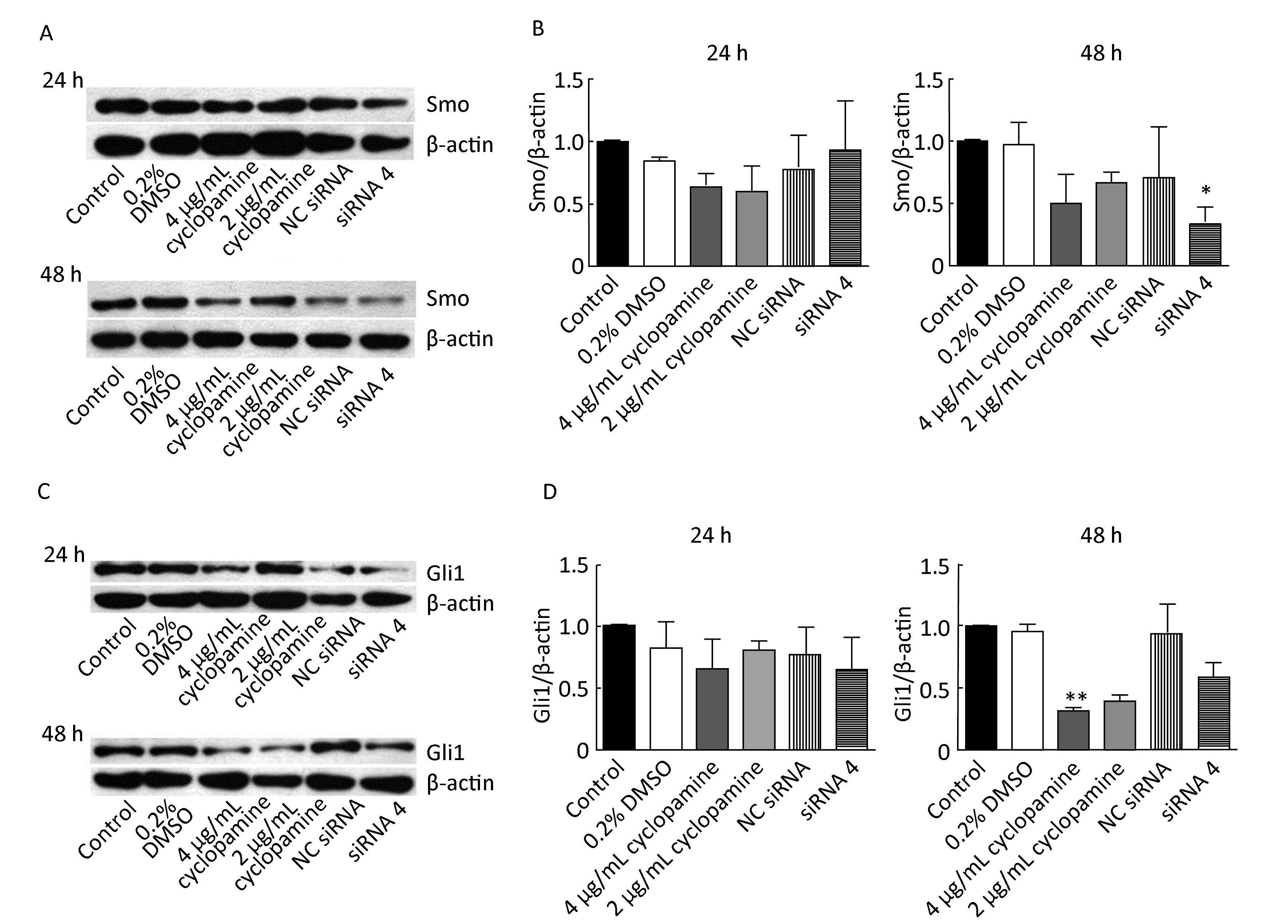
|
Download:
|
| Figure 1 Effect of siRNA 4 and cyclopamine on Smo and Gli1 protein expression at 24 h and 48 h after transfection. (A) Western blot analysis displays Smo protein levels of different groups at 24 h and 48 h after transfection. (B) Results of gray value analysis of Smo protein are shown. (C) Western blot analysis displays Gli1 protein levels of different groups at 24 h and 48 h after transfection. (D) Results of gray value analysis of Gli1 protein are shown. β-actin was used as a loading control. P<0.05 compared to blank control group. **P<0.01 compared to blank control group. | |
Compared with blank control group, Gli1 level of each of the remaining groups at 24 h after transfection was not significantly different (P>0.05), but it was 34.9% and 34.6% lower in siRNA 4 group and 4 μg/mL cyclopamine group, respectively (P>0.05). At 48 h after transfection, Gli1 levels of 4 μg/mL cyclopamine group, 2 μg/mL cyclopamine group, and siRNA 4 group were, respectively, 68.9% (P<0.01), 61.5% (P>0.05), and 42.2% (P>0.05) lower than that of blank control group. Results of Gli2 expression are shown in Figure 2 (A, B). In comparison with blank control group, each of the remaining groups did not exhibit significant difference in Gli2 levels at 24 h or 48 h after transfection. Gli2 levels of siRNA 4 group and 4 μg/mL cyclopamine group at 48 h after transfection
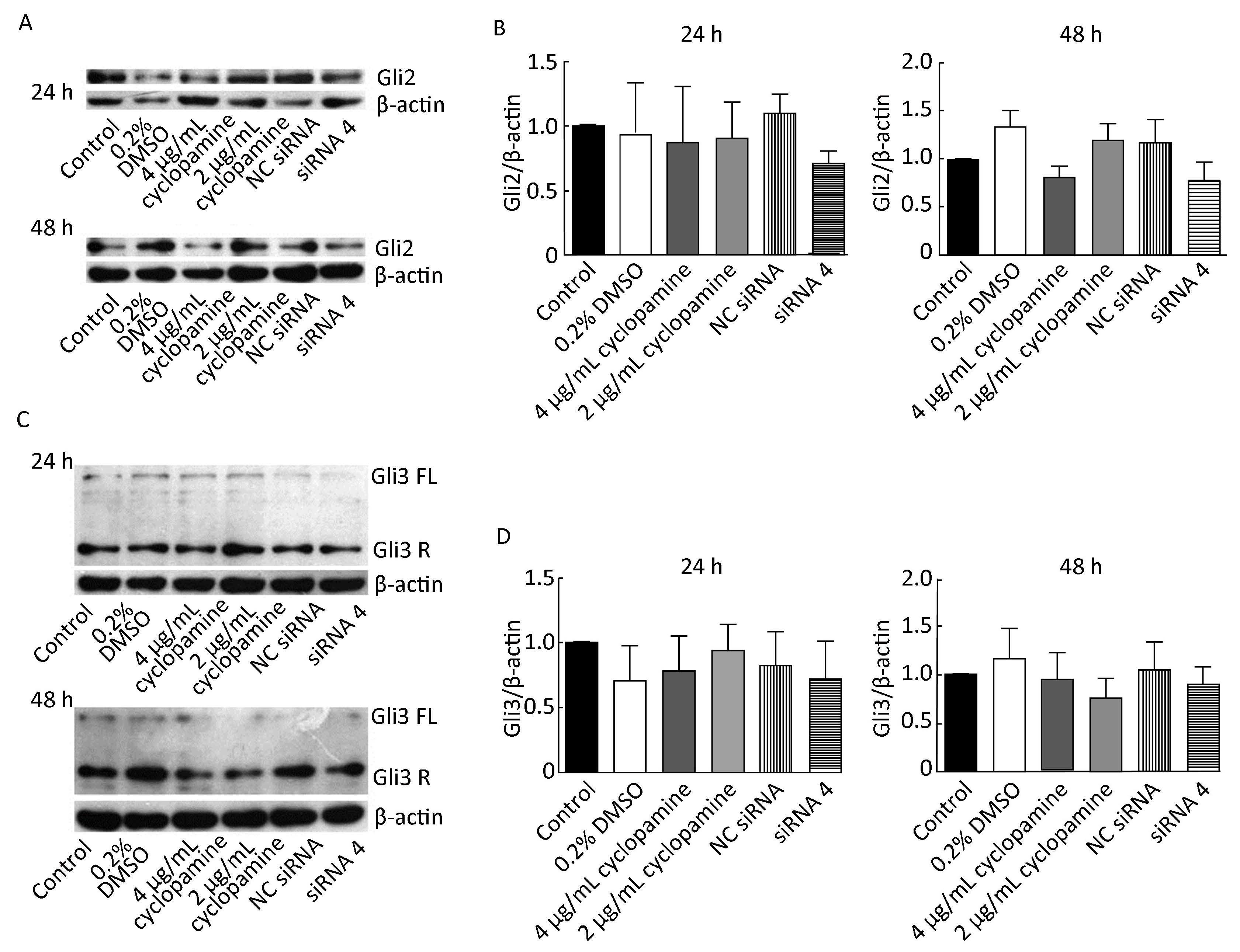
|
Download:
|
| Figure 2 Effect of siRNA 4 and cyclopamine on Gli2 and Gli3 protein expression at 24 h and 48 h after transfection. (A) Western blot analysis displays Gli2 protein levels of different groups at 24 h and 48 h after transfection. (B) Results of gray value analysis of Gli2 protein are shown. (C) Western blot analysis displays Gli3 protein levels of different groups at 24 h and 48 h after transfection. (D) Results of gray value analysis of Gli3 protein (repressor form) are shown. β-actin was used as a loading control. | |
were 23.3% and 19.9% lower than that of blank control group, respectively (P>0.05). Gli3 protein had two forms of fragments, full-length form and repressor form. Due to the unsharpness of the full-length form, gray value analysis was carried out only for the repressor form. As shown in Figure 2 (C, D), there was no obvious difference in Gli3 levels between blank control group and each of the remaining groups at 24 h or 48 h after transfection (P>0.05).
As principal effectors of the hedgehog signaling pathway, Gli transcription factors act as transcriptional activators in their full-length pattern, but they transform to transcriptional repressors when they are degraded by proteasome at C-terminal[8]. Gli1 exists only as transcriptional activator since it maintains in full-length form, whereas Gli2 and Gli3 function as both transcriptional activators and repressors, probably owing to their special structure at C-terminal[1]. As a target gene of hedgehog pathway, Gli1 normally contributes to propagation of hedgehog signaling once it is expressed and thus could be an indicator for hedgehog signaling activity[9]. In this study, siRNA 4 successfully decreased Smo expression but just showed a trend of suppressing Gli1 expression, indicating that hedgehog signaling was not seriously inhibited, probably owing to the insufficient inhibition effect on Smo expresssion. This was closely related to factors such as the limitation of RNAi, siRNA concentration, and function time, which may need further studies. This insufficient inhibition effect of siRNA 4 on Smo expression may also contribute to its nonobvious influence on Gli2 and Gli3 expression and palatal fusion.
Two different doses of cyclopamine groups were set up, namely 2 μg/mL (4.86 μmol/L) and 4 μg/mL (9.72 μmol/L). In comparison with the blank control group, Gli1 expression of 4 μg/mL cyclopamine group was significantly lower, while that of 2 μg/mL
cyclopamine group was not. The results were consistent with previous studies, suggesting that the inhibition effect of cyclopamine on hedgehog signaling was dose dependent and obvious at 10 μmol/L.
In conclusion, Smo siRNA blocked the signaling of hedgehog pathway, which resulted in decreased palatal fusion rate. The mechanism might involve downregulation of transmembrane protein Smo and downstream transcription factor Gli1 expression in the hedgehog pathway.
| 1. | Ryan KE, Chiang C. Hedgehog secretion and signal transduction in vertebrates[J]. JBC Papers in Press , 2012, 287 :17905–13. |
| 2. | Gritli-Linde A. Molecular control of secondary palate development[J]. Dev Bio , 2007, 301 :309–26. doi:10.1016/j.ydbio.2006.07.042 |
| 3. | Rice R, Spencer-Dene B, Connor EC, et al. Disruption of Fgf10/Fgfr2b-coordinated epithelial-mesenchymal interactions causes cleft palate[J]. J Clin Invest , 2004, 113 :1692–700. doi:10.1172/JCI20384 |
| 4. | Lan Y, Jiang R. Sonic hedgehog signaling regulates reciprocal epithelial-mesenchymal interactions controlling palatal outgrowth[J]. Development , 2009, 136 :1387–96. doi:10.1242/dev.028167 |
| 5. | Shimizu N, Aoyama H, Hatakenaka N, et al. An in vitro screening system for characterizing the cleft palate-inducing potential of chemicals and underlying mechanisms[J]. Reprod Toxicol , 2001, 15 :665–72. doi:10.1016/S0890-6238(01)00175-7 |
| 6. | Horn A, Kireva T, Palumbo-Zerr K, et al. Inhibition of hedgehog signaling prevents experimental fibrosis and induces regression of established fibrosis[J]. Ann Rheum Dis , 2012, 71 :785–9. doi:10.1136/annrheumdis-2011-200883 |
| 7. | Chen JK, Taipale J, Cooper MK, et al. Inhibition of Hedgehog signaling by direct binding of cyclopamine to Smoothened[J]. Genes Dev , 2002, 16 :2743–8. doi:10.1101/gad.1025302 |
| 8. | RuiziAltaba A, Sanchez P, Dahmane N. Gli and hedgehog in cancer:tumors, embryos and stem cells[J]. Nat Rev Cancer , 2002, 2 :361–72. doi:10.1038/nrc796 |
| 9. | Bai CB, Auerbach W, Lee JS, et al. Gli2, but not Gli1, is required for initial Shh signaling and ectopic activation of the Shh pathway[J]. Development , 2002, 129 :4753–61. |
 2016, Vol. 29
2016, Vol. 29


